Silicified microcrystalline cellulose (SMCC) is a high-performance pharmaceutical excipient designed for direct compression tablets.
✔ Superior flowability vs MCC, Improved tablet hardness ,Ideal for high-speed tablet production
We are a professional Silicified Microcrystalline Cellulose excipient (SMCC) supplier and manufacturer, providing pharmaceutical-grade excipients for tablet formulation, direct compression systems, and solid dosage development worldwide.In addition,we also supply microcrystalline cellulose (MCC) and colloidal MCC gel for food and pharmaceutical applications.
This page covers not just our supply capabilities — it provides the technical depth that formulation scientists and procurement teams need to make confident decisions about Silicified Microcrystalline Cellulose grade selection, dosage, and application.
👉 Request SMCC datasheet, CoA, or sample
This tablet excipient (SMCC) is a co-processed pharmaceutical excipient produced by intimately combining microcrystalline cellulose (MCC) with a precisely controlled amount of colloidal silicon dioxide (typically 2% w/w). Unlike simple physical blends, the co-processing method creates a composite structure where silicon dioxide particles are embedded within and on the surface of MCC particles at the manufacturing stage.
This structural integration — not just surface coating — is what distinguishes Silicified Microcrystalline Cellulose from a manually blended MCC/SiO₂ mixture. The result is a material with fundamentally different powder behavior: superior flow, enhanced compressibility, and improved batch-to-batch consistency that a post-hoc blend cannot reliably replicate.
Silicified Microcrystalline Cellulose is listed in major pharmacopoeias including USP-NF and Ph. Eur., and is widely regarded as the benchmark co-processed excipient for direct compression tableting.
Key physicochemical characteristics:
We are a professional:

To understand why SMCC outperforms standard MCC in direct compression, it is necessary to understand how powder flow and compressibility work at the particle level.
Microcrystalline cellulose is an excellent compressible excipient, but its flowability is limited by inter-particle cohesion — hydrogen bonding between hydroxyl groups on adjacent cellulose surfaces creates adhesive forces that resist free flow, especially at fine particle sizes.
Colloidal silicon dioxide (fumed silica, ~7–40 nm primary particle size) functions as a glidant by physically separating MCC particles and disrupting these cohesive interactions. When integrated through co-processing, the silica forms a partial coating on the cellulose surface, occupying the most active binding sites and reducing effective inter-particle contact area.
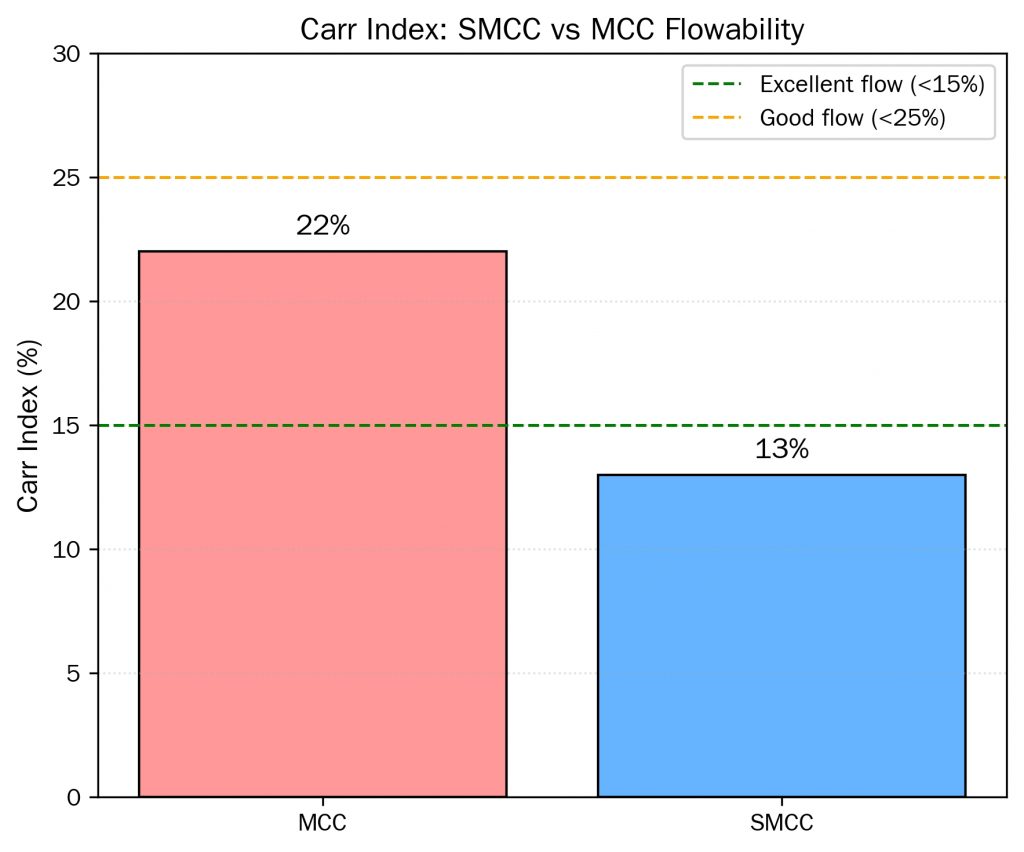
In addition,the outcome is measurable: Carr Index values for SMCC typically fall below 15 (indicating good-to-excellent flow), compared to 20–30+ for standard MCC grades of similar particle size. This difference is critical in high-speed rotary tablet presses, where consistent die filling depends on reliable powder flow from the hopper through the feed frame.
A common assumption is that adding a hard, non-compressible material like silica would reduce tablet hardness. In Silicified Microcrystalline Cellulose, the opposite is observed. Because the silica reduces inter-particle friction, particles rearrange more efficiently during compression, leading to higher packing density and greater surface contact area under the punch force.
Additionally, the reduced internal stress in Silicified Microcrystalline Cellulose tablets (compared to over-lubricated MCC tablets) means that the plastic deformation capacity of cellulose is more fully utilized. Studies have demonstrated that SMCC tablets achieve higher hardness at equivalent compression force, and maintain higher hardness in the presence of magnesium stearate — a critical advantage, as lubricant sensitivity is one of the main failure modes in direct compression formulation.
This tablet excipient (Silicified Microcrystalline Cellulose) is a co-processed excipient produced by co-drying microcrystalline cellulose with colloidal silicon dioxide. Unlike a simple physical blend, the silicon dioxide integrates into the MCC particle surface during processing — and that structural difference drives real performance advantages.
The most significant benefit is flowability. Standard MCC grades, particularly PH-101, can exhibit poor flow in high-speed tablet press hoppers, leading to weight variation and content uniformity issues. The surface-integrated silicon dioxide in Silicified Microcrystalline Cellulose reduces interparticle friction substantially, producing better flow without sacrificing compressibility.
In addition,SMCC is also more resistant to over-lubrication by magnesium stearate. Over-lubrication reduces tablet hardness and slows disintegration — Silicified Microcrystalline Cellulose gives manufacturers a wider processing window and more consistent results at high production speeds.
For formulators working with poorly compressible APIs or running high-speed direct compression processes, SMCC is a technically justified upgrade over standard MCC with well-documented performance advantages.
Silicified microcrystalline cellulose offers several advantages in modern tablet production.
The integrated silicon dioxide reduces inter-particle friction and improves flow through hoppers and feed frames. Consequently, manufacturers achieve more uniform die filling and better tablet weight consistency.
Silicified MCC retains the plastic deformation properties of standard MCC while providing higher compactibility. Therefore, formulators can often achieve target tablet hardness at lower compression forces.
For low-dose formulations, powder segregation can create significant content variability. However, the improved flow and particle interaction of silicified MCC help maintain blend homogeneity throughout the compression process.
Because silicified MCC combines excellent flow and binding performance, manufacturers often reduce the need for additional flow aids or processing steps. As a result, production efficiency improves and formulation complexity decreases.
Consequently, many pharmaceutical companies prefer silicified MCC for direct compression tablets because it supports high-speed manufacturing while maintaining tablet quality and mechanical strength.
The choice between Silicified Microcrystalline Cellulose and standard MCC is not simply a matter of “better vs. worse” — it depends on the specific challenges of the formulation. Below is a technical comparison across the parameters that matter most in tablet development.Compared with standard MCC excipients, Silicified Microcrystalline Cellulose provides better flowability and compressibility.
| Parameter | SMCC | Standard MCC (e.g., PH-102) |
|---|---|---|
| Flowability (Carr Index) | ≤15 (Good–Excellent) | 20–30 (Fair–Poor) |
| Compressibility | Higher tablet hardness per force unit | Good, but lower than SMCC |
| Lubricant sensitivity | Low — maintains hardness with MgSt | Moderate — hardness drops with over-lubrication |
| Content uniformity (low-dose APIs) | Excellent — reduced segregation | Good — may require extra blending steps |
| High-speed tableting suitability | High — reliable die filling | Moderate — may require flow aids |
| Moisture sensitivity | Similar to MCC | Standard baseline |
| Disintegration | Good (comparable to MCC) | Good |
| Regulatory status | USP/NF, Ph. Eur. | USP/NF, Ph. Eur., JP |
| Cost | Slightly higher than standard MCC | Lower baseline cost |
Practical conclusion: If your formulation involves high-speed tableting, a low-dose API requiring tight content uniformity, or a blend that has shown lubricant sensitivity, therefore,silicified microcrystalline celluloseoffers a significant technical advantage over standard MCC. For simpler formulations with adequate flow, standard MCC remains cost-effective.
👉 Related: Colloidal Microcrystalline Cellulose (MCC Gel)
👉 Related: Microcrystalline Cellulose (MCC) Supplier
Both SMCC and standard MCC are widely used pharmaceutical excipients, and for many formulations, conventional MCC performs perfectly well. However, there are specific situations where SMCC delivers measurably better results.
First of all, flowability is the most common reason. Standard MCC — especially fine grades like PH-101 — can be cohesive and flow poorly in high-speed tablet press feed frames, thus causing weight variation and content uniformity problems. In contrast, SMCC’s surface-integrated silicon dioxide significantly reduces interparticle cohesion, improving flow without any loss of compressibility.
Secondly, lubrication sensitivity is the second reason. Specifically, magnesium stearate over-lubrication is a well-known risk in direct compression — excessive mixing reduces tablet hardness and slows disintegration. As a result, SMCC is substantially more resistant to this effect, giving manufacturers a wider and more forgiving processing window.
Finally, SMCC performs better in formulations with high proportions of poorly compressible APIs. For example, when the active ingredient contributes little binding strength, SMCC’s superior compactibility helps maintain tablet hardness at acceptable compression forces.
In short: if your formulation runs well on standard MCC, there is no need to switch. However, if you are experiencing flow problems, lubrication sensitivity, or compactibility challenges with difficult APIs — then SMCC is the logical next step.
Silicified Microcrystalline Cellulose used in pharmaceutical applications should comply with the requirements of the European Pharmacopoeia (Ph Eur), as well as other major pharmacopeial standards when applicable.
Ph Eur specifications typically cover:
Furthermore, pharmaceutical manufacturers commonly require full batch traceability, Certificates of Analysis (COA), and GMP-compliant production documentation.
High-quality silicified MCC suppliers also provide support documentation such as:
Therefore, selecting a supplier that consistently meets Ph Eur requirements helps pharmaceutical companies maintain regulatory compliance and ensure reliable tablet manufacturing performance.According to the European Pharmacopoeia (Ph. Eur.),FDA, pharmaceutical excipients must meet strict quality and purity requirements.
SMCC is commercially available in multiple particle size grades, analogous to the MCC PH-101/PH-102/PH-200 range. Grade selection primarily affects flow rate, bulk density, and suitability for different tableting equipment.
| SMCC Grade | Mean Particle Size | Typical Application |
|---|---|---|
| SMCC 50 | ~50 µm | Formulations requiring finer particle size, blending with fine API powders, improved content uniformity |
| SMCC 90 | ~90 µm | General direct compression, standard tablet presses, balanced flow and compressibility |
Therefore, grade selection should be confirmed through bulk density measurement, flow testing (Carr Index / Hausner Ratio), and compaction simulation (e.g., Heckel analysis or compaction simulator) at the formulation development stage.
SMCC is typically used at 20%–80% by weight of the total tablet blend in direct compression systems. The optimal level depends on the API loading, required tablet hardness, and disintegration target. For high-drug-load formulations (API > 50%), SMCC at 20–30% can still provide meaningful flowability improvement. For low-API formulations, SMCC serves as the primary matrix former at 50–80%.
SMCC is generally compatible with most pharmaceutical excipients. Key considerations:
For a standard direct compression blend using SMCC:

SMCC is used across a broad range of solid dosage formats and therapeutic categories:
As an established SMCC excipient manufacturer and supplier, we provide more than material supply — we support your formulation from development through commercial scale.
| Parameter | Specification |
|---|---|
| Product Name | Silicified Microcrystalline Cellulose (SMCC) |
| Appearance | White to off-white powder |
| SiO₂ Content | ~2% w/w (co-processed) |
| Loss on Drying | ≤5.0% |
| pH (1:10 suspension) | 5.0–7.5 |
| Heavy Metals | ≤10 ppm |
| Microbial Limits | Per USP/Ph. Eur. pharmaceutical excipient standards |
| Available Grades | SMCC 50, SMCC 90 |
| Pharmacopoeial Status | USP/NF, Ph. Eur. |
| Packaging | 25 kg fiber drums, PE inner bag |
| Storage | Cool, dry conditions; protect from moisture |
A physical blend simply mixes the two materials together; the silica does not integrate into the MCC particle structure. Specifically, co-processed SMCC embeds silicon dioxide within the MCC matrix during manufacturing, producing a homogeneous composite particle. Physical blends are less consistent, tend to segregate, and do not deliver the same flowability or compressibility improvements. This is why SMCC is a defined compendial excipient and physical blends are not considered equivalent for regulatory purposes.
SMCC has a similar equilibrium moisture content to standard MCC (~3–5% under typical warehouse conditions). For moisture-sensitive APIs, appropriate packaging (sealed HDPE containers, silica desiccant inserts) and controlled storage conditions are required — the same precautions apply to standard MCC-based formulations. A specific compatibility study between the API and SMCC is recommended during development.
Substituting SMCC for MCC in an existing approved formulation typically constitutes a formulation change that requires regulatory notification or prior approval depending on the market and change classification. Therefore, from a technical standpoint, the compression and flow behavior will differ — in most cases favorably — but compaction force, tablet hardness, and dissolution profiles should still be re-verified.
For most standard tablet formulations using direct compression, SMCC 90 (mean ~90 µm) is the starting point. If your API is very fine (<20 µm) and content uniformity is a concern, SMCC 50 may reduce segregation risk due to closer particle size matching. Our technical team can assist with grade selection based on your formulation brief.
We are both. We manufacture this tablet excipient in-house and supply directly to pharmaceutical manufacturers and contract development organizations (CDMOs) globally. Direct manufacturing means we control quality at every step and can respond quickly to supply or specification queries.
We provide full Certificate of Analysis (CoA), Material Safety Data Sheet (MSDS/SDS), DMF reference support where applicable, and technical data packages for regulatory filing support. Please contact us to discuss your specific documentation requirements.
Formulators mainly use SMCC as a direct compression excipient in tablet formulation, improving flowability, compressibility, and content uniformity.
Leading SMCC suppliers provide pharmaceutical-grade material with USP/NF and Ph. Eur. compliance, consistent particle size distribution, and technical support.
Looking for a reliable silicified microcrystalline cellulose supplier with consistent quality and fast delivery?