Microcrystalline cellulose supplier providing MCC powder E460(i) for food, pharmaceutical, and industrial applications. This microcrystalline cellulose excipient is widely used for tablet binding, suspension stability, and fat replacement systems.According to FDA regulations, microcrystalline cellulose is recognized as safe for use in food and pharmaceutical applications. FDA guidelines
✔ Food grade & pharma grade MCC ✔ Stable bulk supply ✔ Technical formulation support
[Request a Quote] | [Get Free Sample]
Related solutions: colloidal MCC stabilizer | HPMC thickener | pharmaceutical excipient solutions
Microcrystalline cellulose E460(i) is widely used as a colloidal MCC stabilizer in beverage and dairy systems, and as a core material in pharmaceutical excipient solutions.
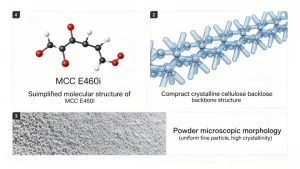
Microcrystalline cellulose (MCC) is a purified, partially depolymerized cellulose derived from natural plant fibers such as wood pulp. It is widely used as a microcrystalline cellulose excipient due to its excellent compressibility, stability, and safety profile.
Produced through partial acid hydrolysis of alpha-cellulose, MCC retains only the highly ordered crystalline regions of the cellulose chain. The result is a white, free-flowing powder with a crystallinity index of 55–80%, a degree of polymerization of 150–300, and particle sizes ranging from 20 to 200 micrometers depending on grade.
This structural purity is what separates microcrystalline cellulose from other cellulose materials. E460(ii) (powdered cellulose) is mechanically ground without hydrolysis and lacks the crystalline structure that gives MCC its functional properties. E466 (CMC) is a chemically modified, water-soluble derivative. MCC is neither — it is purified native cellulose in its most functional form, and that distinction drives everything about how it performs.
Microcrystalline cellulose carries no ADI (Acceptable Daily Intake) restriction from JECFA, is listed as GRAS by the FDA under 21 CFR 182.90, and is approved under EU Regulation (EC) No 1333/2008 as E460(i). It is listed in USP-NF, EP, JP, and BP pharmacopeias. It contributes zero calories, is not absorbed by the human body, and is suitable for vegan, halal, and kosher applications.
The global adoption of microcrystalline cellulose across food and pharmaceutical manufacturing is not accidental. MCC delivers performance that most alternative ingredients cannot replicate — and it does so across multiple application categories simultaneously.
In tablet manufacturing, the crystalline structure of microcrystalline cellulose gives it exceptional plastic deformation behavior under compressive force. When pressure is applied in a tablet press, MCC particles deform and interlock, forming strong mechanical bonds that hold the tablet together without requiring high compression forces or wet granulation.
This makes microcrystalline cellulose excipient the standard choice for direct compression manufacturing — a process that eliminates wet granulation, reduces production steps, lowers energy consumption, and is particularly critical for moisture-sensitive or heat-sensitive active pharmaceutical ingredients.
Beyond binding, MCC functions simultaneously as a disintegrant. Upon contact with gastrointestinal fluid, MCC particles absorb water rapidly and swell, generating internal stress that breaks the tablet apart and releases the API. This dual function — binder during compression, disintegrant upon dissolution — is unique to MCC and represents a significant formulation advantage.
Different pharmaceutical grades serve different manufacturing requirements. PH-101 (50 μm average particle size) is the standard direct compression grade. PH-102 (100 μm) offers improved flowability for high-speed rotary presses. PH-200 (180 μm) is optimized for very high-speed production with maximum flow. PH-301 and PH-302 are dense grades for applications requiring higher bulk density. Grade selection has a direct and measurable impact on tablet hardness, friability, disintegration time, and press throughput — and must be evaluated under actual manufacturing conditions.
When microcrystalline cellulose is properly dispersed in water, it does not dissolve — it forms a three-dimensional particle network through hydrogen bonding between individual MCC particles. This network has a measurable yield stress, typically 0.5 to 5 Pa depending on concentration and grade. Below this yield stress, the network behaves as a solid structure, holding suspended particles — cocoa, minerals, protein, fruit pulp, fiber — in place against gravitational settling.
This is a fundamentally different mechanism from viscosity-building hydrocolloids like xanthan gum or guar gum. Those ingredients slow settling by increasing the resistance to particle movement through the liquid medium. MCC prevents settling by creating a physical structure that eliminates the driving force for movement altogether. The result is significantly more robust suspension stability, particularly for long shelf-life products and challenging particle loads.
Used together with colloidal MCC stabilizer systems, microcrystalline cellulose achieves full network formation in liquid applications. In colloidal MCC, MCC is co-processed with CMC at a typical ratio of 85:15 to 90:10. The CMC acts as a dispersant, separating individual MCC particles during hydration and allowing the network to form completely. Standard MCC powder without CMC will not adequately disperse in water and will not develop a functional suspension network.

The MCC network is also thixotropic — it breaks down under shear during mixing, pumping, or pouring, then rebuilds at rest. For manufacturers, this means low viscosity during processing and reliable stability during storage and distribution.
Microcrystalline cellulose is one of the most effective fat replacers available for food formulators. In low-fat and reduced-calorie products, the removal of fat creates textural deficiencies — loss of body, mouthfeel, lubricity, and emulsion stability — that are difficult to compensate for with other ingredients.
MCC addresses these deficiencies through two mechanisms. First, the particle network created by dispersed MCC mimics the physical texture of fat droplets in an emulsion, providing body and mouthfeel without caloric contribution. Second, MCC particles adsorb at the oil-water interface in emulsified systems, physically stabilizing the emulsion against coalescence and phase separation.
This makes microcrystalline cellulose a primary tool in fat-reduced dressings, dairy products, sauces, spreads, and frozen desserts. Use levels typically range from 0.5% to 1.5% depending on the target fat reduction and desired texture profile.
Microcrystalline cellulose serves as a critical functional ingredient across a broad range of food and pharmaceutical applications. Each application category places different demands on MCC grade selection, concentration, and processing conditions.E460(i) is widely used under EU regulation standards for food additives. EU food additive regulation
Microcrystalline cellulose is widely used in beverage stabilizer and ice cream stabilizer applications, as well as pharmaceutical excipient systems.

Pharmaceutical tablets and capsules. MCC is the most widely used excipient in solid dosage form manufacturing globally. It functions as a direct compression binder, disintegrant, and flow aid across the full range of tablet and capsule formulations.It is a core material in pharmaceutical excipient solutions for tablet, capsule, and controlled-release systems.
Pharmaceutical pellets. In extrusion-spheronization, MCC acts as a spheronization aid — producing smooth, dense, spherical pellets with narrow size distribution at loadings of 40%–70% of pellet mass. No other excipient matches MCC performance in this application.

Beverages and nutritional drinks. Colloidal MCC prevents sedimentation of cocoa, minerals, protein, and fiber in chocolate milk, nutritional shakes, plant-based beverages, and fortified drinks. Typical use levels are 0.3%–1.2%.

Ice cream and frozen desserts. MCC controls ice crystal growth during freeze-thaw cycling, maintaining smooth texture throughout shelf life. Typical use levels are 0.2%–0.5%.
Sauces, dressings, and emulsions. MCC stabilizes oil-water emulsions and provides fat-like texture in reduced-fat formulations at 0.5%–1.5%.
Bakery and dry mixes. MCC improves batter viscosity and crumb structure in gluten-free bakery and functions as an anti-caking agent in powder blends.
Understanding the differences between these three cellulose-based ingredients is essential for correct formulation decisions. They are frequently confused, frequently misapplied, and frequently used together — but their mechanisms are entirely different.
Microcrystalline cellulose (MCC / E460(i)) is insoluble in water. It functions by forming a physical particle network with yield stress. It provides structural stabilization, fat replacement, and tablet compressibility. It contributes zero calories and requires no chemical modification.
Compared with CMC, microcrystalline cellulose does not dissolve but forms a physical network. For viscosity control, you may also explore our CMC supplier page or HPMC supplier solutions.
CMC (carboxymethyl cellulose / E466) is water-soluble. It functions by increasing solution viscosity through polymer chain entanglement. It provides thickening and gelling but does not create a yield-stress network. It is frequently co-processed with MCC (as colloidal MCC) to assist MCC dispersion and add viscosity contribution.
HPMC (hydroxypropyl methylcellulose / E464) is water-soluble and thermally gelling. It dissolves in cold water and gels upon heating — the opposite of most hydrocolloids. It is widely used in pharmaceutical film coating, controlled-release matrix tablets, vegetarian capsules, and food applications requiring thermal gelation such as restructured meat and gluten-free bakery. For viscosity-critical applications, our HPMC thickener page provides full grade specifications.
The three ingredients are complementary rather than interchangeable. Many high-performance formulations use MCC and CMC together for suspension stability, or MCC and HPMC together for combined structural and viscosity control in pharmaceutical or food systems.
Looking to buy microcrystalline cellulose powder in bulk? We supply food-grade and pharmaceutical-grade MCC with consistent quality, full regulatory documentation, and global delivery capability.As a professional microcrystalline cellulose supplier, we provide bulk MCC powder with stable quality and global delivery.
Our microcrystalline cellulose product range includes:
Every batch is tested against USP-NF, EP, and Codex Alimentarius specifications for particle size, moisture content, pH, bulk density, ash content, and microbiological compliance. We provide full traceability from raw material to finished product.
[Request a Quote] | [Get Free Sample] | [Download Technical Datasheet]
Microcrystalline cellulose is used as a binder, stabilizer, disintegrant, and microcrystalline cellulose excipient in food and pharmaceutical applications. In food, it stabilizes suspensions, replaces fat, and controls ice crystal growth. In pharmaceuticals, it enables direct compression tablet manufacturing and pellet spheronization.
Colloidal MCC stabilizer is a co-processed system of microcrystalline cellulose (E460i) and CMC (E466), typically in an 85:15 ratio. It is specifically designed for liquid applications such as beverages, dairy, and sauces, where standard MCC cannot fully disperse. Learn more about colloidal MCC stabilizer.
Standard pharmaceutical grades include PH-101, PH-102, PH-200, PH-301, and PH-302, each with different particle sizes and bulk densities suited to different manufacturing processes. Food-grade and colloidal MCC grades are also available for beverage, dairy, and emulsion applications.
Standard MCC powder does not fully disperse in water without assistance. Colloidal MCC is co-processed with CMC (typically at 85:15 ratio), which acts as a dispersant to ensure complete hydration and network formation in liquid systems. Colloidal MCC is required for effective suspension stability in beverages and dairy.
Yes. MCC (E460i) is approved by the FDA (GRAS), EFSA, and JECFA with no ADI restriction. It is listed in all major pharmacopeias and has a long history of safe use in food and pharmaceutical products globally.
For suspension stability in beverages, typical use levels range from 0.3% to 1.2% of finished product weight, depending on particle load, target shelf life, and processing conditions.
Yes. Microcrystalline cellulose is widely used in reduced-fat and low-calorie formulations to replicate the texture and mouthfeel of fat. It contributes zero calories and is particularly effective in dressings, dairy products, frozen desserts, and spreads at use levels of 0.5%–1.5%.
Yes. Microcrystalline cellulose is derived entirely from plant-based cellulose (wood pulp or cotton linters). It contains no animal-derived ingredients and is suitable for vegan, vegetarian, halal, and kosher formulations.
You can buy MCC powder from a reliable microcrystalline cellulose supplier offering food and pharma grade materials.
For technical specifications, regulatory documents, sample requests, and bulk pricing on food-grade and pharmaceutical-grade microcrystalline cellulose, contact our technical sales team.
Reviews
There are no reviews yet.