As a trusted microcrystalline cellulose manufacturer and bulk supplier, we provide pharmaceutical-grade, food-grade, and industrial-grade MCC to clients worldwide. Therefore,in the context of the Microcrystalline Cellulose in Pharmaceutical Industry, our material is widely used in tablet binding, drug formulation, and controlled-release systems.
From crystal structure to production-ready formulations — a rigorous technical reference covering microcrystalline cellulose compression science, grade selection, dosage form applications, SMCC comparison, compatibility risks, and quality control specifications.
Key Takeaways

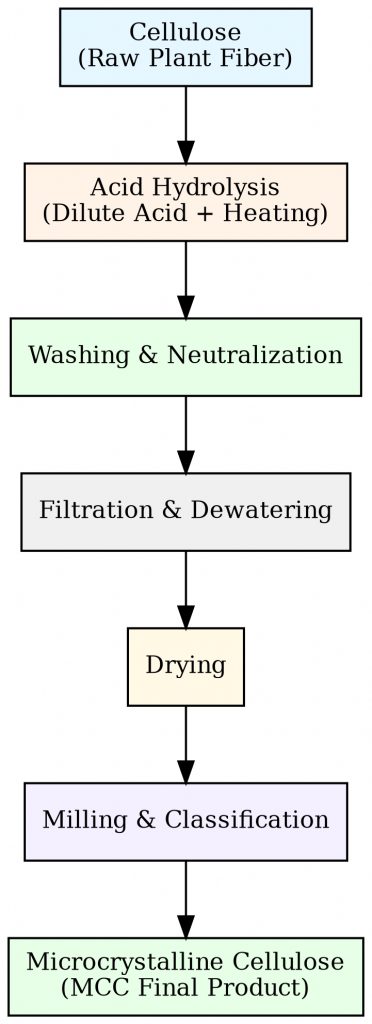
Microcrystalline cellulose (MCC) is a purified, partially depolymerized form of cellulose derived from natural plant sources — primarily wood pulp with an alpha-cellulose content of ≥99%, or pharmaceutical-grade cotton linters. The manufacturing process centers on controlled acid hydrolysis: dilute hydrochloric acid (typically 2.5 mol/L) selectively cleaves the amorphous regions of cellulose chains at defined temperature and reaction time, leaving behind the thermodynamically stable crystalline domains intact. Meanwhile,the resulting suspension is then washed, neutralized, and spray-dried into the familiar white, free-flowing powder used in tablet manufacturing.
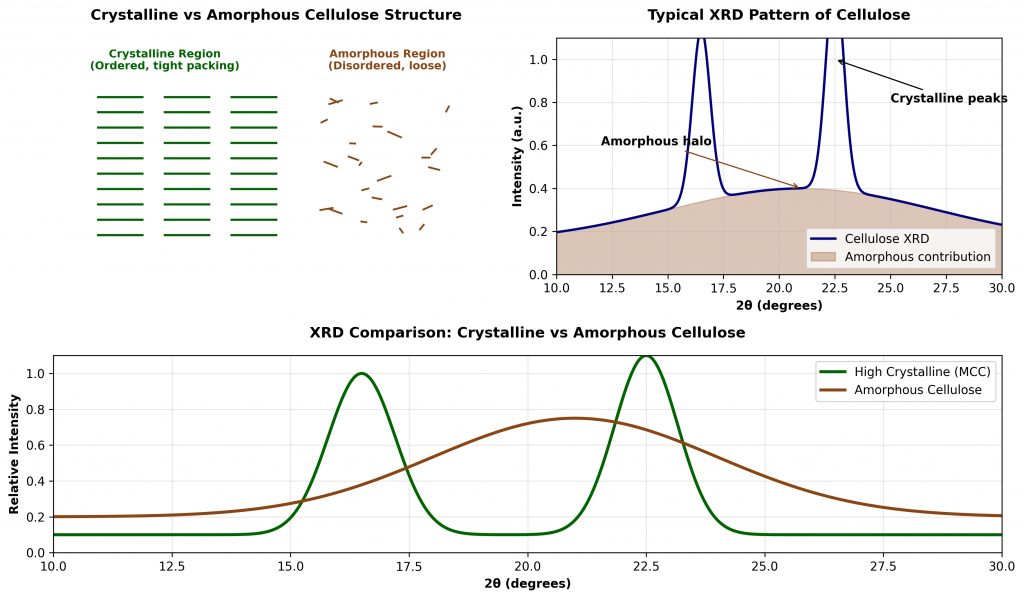
Therefore,This process yields a material with a crystallinity index of 70–80%, considerably higher than native cellulose (~50–60%). That elevated crystalline order is the molecular foundation of MCC’s exceptional compressibility — it determines how the material responds under tablet press forces at the nanoscale.
🌿
Primarily sourced from softwood pulp (α-cellulose ≥99%) or cotton linters, ensuring high purity, low heavy metals, and batch-to-batch consistency essential for pharmaceutical use.
⚗️
Dilute HCl hydrolysis removes amorphous cellulose regions, retaining crystalline segments at the Leveling-Off Degree of Polymerization (LODP). Spray drying determines final particle morphology.
📐
Cellulose type I crystal lattice; LODP of approximately 150–250 glucose units; high specific surface area of 1–2 m²/g supporting both adsorption and capillary wetting functions.
🏭
Available as spray-dried powder (standard), agglomerated/granular grades for improved flow, and co-processed forms including silicified MCC (SMCC) and MCC-mannitol blends for ODT applications.
pharmaceutical excipient MCC holds a unique regulatory position: it is simultaneously classified as GRAS (Generally Recognized As Safe) by the U.S. FDA with no established Acceptable Daily Intake (ADI), and formally monographed across all major global pharmacopeias.
| Pharmacopeia | Monograph Name | Key Specifications | Region |
|---|---|---|---|
| USP/NF | Microcrystalline Cellulose | pH 5.0–7.5; moisture ≤5.0%; ash ≤0.1% | United States |
| Ph. Eur. 11.0 | Cellulosum microcristallinum | Equivalent to USP; additional heavy metals limits | European Union |
| JP 18 | Microcrystalline Cellulose | Specific limits for particle size distribution and whiteness | Japan |
| ChP 2020 | Microcrystalline Cellulose | Aligned with USP; supplementary local regulatory clauses | China |
The absence of an ADI upper limit is practically significant: formulators can use pharmaceutical excipient pharmaceutical excipient MCC at levels up to 90% of tablet weight without triggering any regulatory dose-restriction discussion, a freedom unavailable with many synthetic binders.
Microcrystalline Cellulose in Pharmaceutical Industry applications mainly centers on tablet manufacturing. Therefore,Pharmaceutical formulators choose MCC because it combines excellent binding properties, flowability, and disintegration performance in a single excipient.
Unlike many traditional binders, MCC allows direct compression processing without requiring wet granulation. As a result, manufacturers can simplify production, reduce processing costs, and improve manufacturing efficiency.
Furthermore, MCC exhibits excellent compatibility with a wide range of active pharmaceutical ingredients (APIs). Therefore, formulators frequently select MCC for immediate-release tablets, chewable tablets, and nutraceutical products.
Because MCC provides both mechanical strength and rapid tablet breakup after administration, it remains one of the most widely used pharmaceutical excipients worldwide.
One of the key reasons for the widespread use of Microcrystalline Cellulose in Pharmaceutical Industry formulations is its unique compression behavior.
When compression force is applied during tablet production, MCC particles undergo plastic deformation rather than brittle fracture. Consequently, individual particles flatten and interlock with neighboring particles.
This particle-to-particle bonding creates strong tablets even at relatively low compression forces. In addition, MCC maintains tablet integrity while reducing the need for additional binders.
Moreover, the porous structure of MCC promotes rapid water penetration after administration. Therefore, tablets can quickly disintegrate and release the active ingredient.
This combination of plastic deformation, strong bonding, and efficient disintegration makes MCC one of the most versatile pharmaceutical excipients available today.
Direct compression remains one of the most efficient tablet manufacturing methods, and MCC plays a critical role in this process.
Key benefits include:
Additionally, MCC enables manufacturers to eliminate wet granulation in many formulations. As a result, production becomes faster, cleaner, and more economical.
For pharmaceutical companies seeking reliable excipient performance, MCC continues to be the preferred material for direct compression tablet manufacturing.
Frequently Asked Questions
The most common commercial MCC grade naming follows the Avicel® (FMC BioPolymer) convention, now adopted industry-wide. Each grade differs in mean particle size, bulk density, moisture content, and flowability — properties that collectively determine suitability for specific manufacturing processes.
| Grade | Mean Particle Size (μm) | Bulk Density (g/mL) | Moisture (% max) | Primary Application | Recommended Process |
|---|---|---|---|---|---|
| PH-101 | ~50 | 0.26–0.31 | 5.0 | Wet granulation, small-scale DC | Wet Granulation |
| PH-102 | ~90 | 0.28–0.33 | 5.0 | Standard direct compression | Direct Compression |
| PH-103 | ~50 | 0.26–0.31 | 3.0 | Moisture-sensitive APIs | DC / Wet Gran. |
| PH-105 | ~20 | 0.20–0.30 | 5.0 | ODT mouthfeel, capsule fill | ODT / Capsule |
| PH-112 | ~90 | 0.28–0.33 | 1.5 | Highly hygroscopic APIs, DC | Direct Compression |
| PH-200 | ~180 | 0.28–0.35 | 5.0 | High-speed DC lines, best flow | Direct Compression |
| PH-301 / 302 | ~50 / 90 | 0.34–0.45 | 5.0 | High-density capsule filling | Capsule Fill |
Grade selection should be driven by four core variables:
Related excipients: HPMC, CMC, food stabilizers
Microcrystalline cellulose in the pharmaceutical industry is widely regarded as one of the most effective binders in solid dosage tablet formulation. Its inherent cohesive properties allow direct compression filler to bind active pharmaceutical ingredients and other excipients into a compact, uniform tablet mass under compression. The result is a tablet with strong mechanical integrity — resistant to chipping, capping, and friability during high-speed manufacturing, transportation, and consumer handling. Unlike some synthetic binders, MCC achieves this without compromising the release profile of the active ingredient, making it suitable for a broad range of therapeutic compounds.
One of the most commercially significant roles of microcrystalline cellulose in pharmaceutical industry applications is its function as a direct compression excipient. direct compression filler possesses exceptional compressibility and flowability, enabling tablet manufacturers to compress powder blends directly into tablets without the additional steps of wet or dry granulation. This simplifies the manufacturing process, reduces production time and cost, and minimizes the risk of moisture or heat degradation of sensitive active ingredients. Its consistent particle size and bulk density across production batches further support reliable, reproducible tablet weight and hardness — critical quality parameters in pharmaceutical manufacturing.
The use of microcrystalline cellulose in the pharmaceutical industry extends beyond standard formulations into advanced drug delivery design. When incorporated into a matrix tablet system, direct compression filler works in combination with rate-controlling polymers to regulate the diffusion and dissolution of the active pharmaceutical ingredient over an extended period. This enables formulators to design once-daily or twice-daily dosing regimens, improving patient compliance and maintaining therapeutic drug levels within the desired range. The ability to fine-tune release kinetics by adjusting MCC grade, particle size, and concentration makes it a flexible and reliable component in advanced oral drug delivery systems.
Formulators increasingly rely on microcrystalline cellulose pharmaceutical industry-grade material as an ideal filler and diluent for both hard gelatin and hydroxypropyl methylcellulose (HPMC) capsules. Its low moisture content, excellent flow characteristics, and chemical inertness make it well-suited for filling operations on high-speed encapsulation equipment. direct compression filler provides the bulk volume necessary to achieve consistent fill weights, particularly when the active ingredient is present in a low dose. Its compatibility with a wide range of APIs — including moisture-sensitive and hygroscopic compounds — further broadens its utility as a capsule filler across diverse pharmaceutical product lines.
Among the many roles of microcrystalline cellulose in pharmaceutical industry formulation, its contribution to tablet disintegration performance remains one of the most clinically impactful. Its highly porous structure enables rapid uptake of water upon contact with gastrointestinal fluids, causing the tablet matrix to swell and break apart efficiently. This promotes fast disintegration and facilitates the dissolution of the active pharmaceutical ingredient, directly supporting bioavailability. In formulations where rapid onset of action is clinically important, direct compression filler serves as a reliable disintegration enhancer — ensuring that tablets perform consistently across varying physiological conditions within the patient population.
For formulations containing highly hydrophobic APIs (water contact angle >90°), direct compression filler’s wetting-promoting role is particularly valuable: even where the API itself resists wetting, the microcrystalline cellulosenetwork channels water through the tablet, enabling disintegration to proceed.
In fmulations, Colloidal Microcrystalline Cellulose Gel is widely used in suspension systems and stabilizers.

Microcrystalline cellulose is widely available as a pharmaceutical excipient.
You can check our Microcrystalline Cellulose for detailed specifications, COA, and bulk supply options.
Silicified Microcrystalline Cellulose (SMCC) is produced by co-processing microcrystalline cellulose with approximately 2% colloidal silicon dioxide, resulting in a composite excipient where silica particles are intimately associated with the MCC surface. The two materials share the same appearance and are used in identical dosage forms, yet deliver meaningfully different performance profiles.
General-purpose DC excipient
Recommended Upgrade
High-performance DC excipient
Reference comparator
💡 When to Upgrade from MCC to SMCC
Consider SMCC when: press speed exceeds 80,000 tablets/hour; blended powder Carr Index exceeds 28; API exhibits high electrostatic propensity; or production line experience includes weight variation failures attributed to poor hopper flow. SMCC typically delivers a 30–50% flow improvement without any additional processing steps or equipment changes.
In some controlled-release systems, microcrystalline cellulose is combined with other excipients such as
Hydroxypropyl Methylcellulose and
Carboxymethyl Cellulose to optimize release profiles.
Microcrystalline cellulose (MCC) generally outperforms pregelatinized starch (PGS) as a dry binder in tablet formulations. MCC provides higher binding efficiency, greater dilution potential, and broader compatibility with pharmaceutical active ingredients.
In addition, MCC supports direct compression manufacturing more effectively because of its excellent plastic deformation properties. As a result, formulators often achieve stronger tablets at lower compression forces.
However, pregelatinized starch offers certain advantages. It can improve tablet disintegration and reduce formulation cost. Therefore, many pharmaceutical manufacturers combine MCC and PGS to balance compressibility, disintegration, and economic performance.
For direct compression tablets, MCC remains the preferred primary binder, while PGS often serves as a complementary excipient.
Microcrystalline cellulose is fully plant-derived and suitable for vegan and vegetarian pharmaceutical formulations. Manufacturers typically produce MCC from purified wood pulp or cotton linters without using any animal-derived ingredients.
Furthermore, MCC is naturally gluten-free because it does not originate from wheat, barley, rye, or other gluten-containing grains.
As a result, pharmaceutical companies frequently use MCC in vegan tablets, vegetarian capsules, nutraceutical products, and gluten-free formulations. Nevertheless, manufacturers should obtain supporting documentation and certification from suppliers when formal vegan or gluten-free claims are required.
Compressibility is one of the most important quality parameters for MCC used in pharmaceutical manufacturing.
The most reliable evaluation method involves compressing pure MCC at multiple force levels and measuring tablet hardness. A qualified pharmaceutical-grade MCC typically produces tablets with hardness exceeding 6 kP at approximately 10 kN compression force.
Additionally, formulators often use Heckel analysis to assess plastic deformation behavior. Lower mean yield pressure values generally indicate better compressibility and improved direct compression performance.
For routine quality control, manufacturers should compare incoming lots against established reference standards. Significant variation in compressibility profiles may indicate a supplier consistency issue and should trigger further investigation.
Direct compression is microcrystalline cellulose’s dominant application. The following framework illustrates a workable starting point for a typical API at moderate loading:
Active Pharmaceutical Ingredient400 mg (40%)
microcrystalline cellulose PH-102 (binder/filler)500 mg (50%)
Croscarmellose Sodium (disintegrant)50 mg (5%)
Magnesium Stearate (lubricant)10 mg (1%)
Colloidal Silicon Dioxide (glidant)5 mg (0.5%)
Target Hardness8–12 kP
Critical process note: Magnesium stearate is a hydrophobic lubricant that, when over-blended, forms a thin film on microcrystalline cellulose particle surfaces that blocks capillary wetting and significantly retards disintegration. Lubricant blending time should be rigorously controlled at 3–5 minutes. All other excipients — API, MCC, disintegrant, glidant — should be pre-blended to homogeneity before lubricant addition.
ODT formulations impose conflicting demands: rapid oral disintegration (target <60 seconds per regulatory definition) while maintaining adequate structural integrity for packaging, shipping, and handling. pharmaceutical excipient MCC PH-105 (mean particle ~20 μm) addresses both requirements.
The ultrafine particle size of PH-105 is imperceptible on the tongue and contributes to smooth mouthfeel — a significant patient-experience differentiator in pediatric and geriatric populations. The greater specific surface area also accelerates water penetration and tablet disintegration. In ODT formulations, PH-105 is typically combined with mannitol (for taste and fast dissolution), crospovidone (PVPP, for rapid disintegration), and a flavor/sweetener system.
In hard gelatin or HPMC capsules, pharmaceutical excipient MCC serves primarily as a diluent and flow modifier. The PH-301 and PH-302 grades — with bulk densities of 0.34–0.45 g/mL, considerably higher than standard grades — maximize fill weight per capsule size, reducing formulation cost and capsule count per patient dose.
Automated capsule filling equipment typically requires powder Carr Index <25 to achieve consistent fill weights and avoid bridging at the dosator. PH-302 (mean particle ~90 μm, high bulk density) generally meets this requirement without additional glidant.
Even when the manufacturing process is wet granulation, pharmaceutical excipient MCC is commonly added extragranularly after milling — typically at 10–20% of total formulation weight. This restores the compressibility lost during granulation (where high shear and liquid addition can over-densify particles and reduce their plastic deformation capacity), enabling tablets with acceptable hardness even from poorly compressible granules. This hybrid strategy is particularly effective for APIs where wet granulation is required for content uniformity but the resulting granule is inherently brittle.
MCC’s broad chemical inertness is well established, but formulators should be aware of specific interaction pathways that can compromise product quality under real manufacturing and storage conditions.
| Risk Type | Trigger Conditions | Example APIs Affected | Mitigation Strategy |
|---|---|---|---|
| Moisture transfer to API | High RH storage; MCC moisture >5%; packaging failure | Aspirin, omeprazole, cefaclor | Select PH-112 (≤1.5% moisture); desiccant packaging; WVTR-specified container |
| Oxidation promotion | Trace transition metals in MCC + oxygen-sensitive API | Vitamin C, PUFAs, ranitidine | Specify low heavy-metal grade; add antioxidant (BHT, ascorbic acid); nitrogen purge |
| Cellulose-amine adduct | Elevated temperature (>60°C); primary amine API; acidic pH | Primary amine-containing compounds | Accelerated stability study (40°C/75%RH, 4 weeks) before scale-up; monitor by HPLC |
| Lubricant over-blending | MgSt mixing >10 min with MCC | All direct compression formulations | Strict lubricant blending time control (3–5 min); consider sodium stearyl fumarate as alternative |
The industry-standard approach for confirming API–MCC compatibility before formulation scale-up is a binary compatibility study: blend API with pharmaceutical excipient MCC in a 1:1 ratio, store at 40°C/75%RH and 60°C/ambient humidity for 2–4 weeks, and assay by HPLC for API degradation. Degradation <1% relative to API alone at both conditions is generally considered indicative of acceptable compatibility for further development.
When qualifying MCC from a new supplier or reviewing incoming material, the following parameters should appear in your raw material specification — not just as regulatory checkboxes but because each directly impacts formulation performance.
| Test Parameter | USP Limit | Formulation Impact | Test Method |
|---|---|---|---|
| Loss on Drying (moisture) | ≤5.0% | Directly governs compressibility; each 1% drop in moisture can reduce tablet hardness by 10–20% | Oven drying, 105°C for 3h (or Karl Fischer titration for precision) |
| pH (1% w/v suspension) | 5.0–7.5 | Affects compatibility with pH-sensitive APIs; outside range suggests acid/alkali contamination | pH electrode in aqueous suspension |
| Particle Size Distribution (D50) | Grade-specific (internal ±15%) | Governs flowability, blend uniformity, and compression behavior; batch variation >15% in D50 requires investigation | Laser diffraction (wet or dry) |
| Bulk and Tapped Density | Grade-specific | Determines capsule fill weight, hopper design, and Carr Index (flowability indicator) | USP <616> graduated cylinder method |
| Residue on Ignition (ash) | ≤0.1% | Indicator of inorganic impurities and heavy metal residues that could catalyze API oxidation | Muffle furnace ignition |
| Microbial Limits | TAMC ≤1000 CFU/g | Patient safety compliance; bioburden exceeding limits invalidates batch for pharmaceutical use | USP <62> microbial enumeration |
MCC is listed in major pharmacopeias including USP and Ph. Eur.
(see USP official monograph: https://www.usp.org)
⚠️ Batch Consistency Warning
MCC compressibility is highly sensitive to moisture content: a drop of just 1% in lot moisture can reduce tablet hardness by 10–20%. Inter-lot moisture variation is one of the most common root causes of tablet hardness OOS events. Consider incorporating cellulose-based binder moisture content as a Process Analytical Technology (PAT) monitoring parameter during continuous manufacturing validation, and establish tighter internal limits (e.g., 3.0–4.5%) versus the pharmacopeial maximum of 5.0%.
Yes. MCC is plant-derived, vegan-friendly, and naturally gluten-free. Manufacturers commonly use it in pharmaceutical and nutraceutical formulations.
MCC provides stronger binding, better compressibility, and higher dilution potential. Pregelatinized starch may improve disintegration and reduce formulation cost.
Formulators typically assess tablet hardness, compression profiles, and Heckel analysis results to verify MCC compressibility and batch consistency.
structured as three tiers (sample → small commercial → full commercial) so buyers at any stage can self-identify. Includes the practical advice to use a distributor if annual volume is below 500 kg, which is genuinely useful and builds trust.
deliberately avoids exact numbers (which go stale fast and can anchor negotiations badly) and instead uses a tiered positioning framework: “low single digits / mid single digits / upper single digits / low double digits.” This is honest, defensible, and still gives procurement teams a real budget anchor. It also shows the volume discount logic (FCL, annual contract), which is what B2B buyers actually care about.
separated into stock grades vs. make-to-order, and crucially includes the often-overlooked incoming QC testing window (5–10 days) that procurement teams routinely forget to build into their timelines. The safety stock recommendation (4–8 weeks) is actionable.
laid out as a documentation table rather than a bullet list, because buyers in regulated markets (FDA, EMA, PMDA) need to know which specific documents they can request. The DMF/CEP distinction is the detail that separates a knowledgeable supplier from a generic one in the eyes of a regulatory affairs team.
Our microcrystalline cellulose is available for immediate industrial supply in standard and custom particle size grades. Minimum order quantities (MOQ) start at 25 kg for sample evaluation and scale to full pallet or container loads for ongoing procurement. Detailed specification sheets — including particle size distribution, moisture content, bulk density, and compliance certifications (USP/NF, EP, FCC) — are available upon request. Contact our technical sales team to receive a product datasheet, request a sample, or discuss tailored supply arrangements for your production needs.
In summary, the role of MCC in the Microcrystalline Cellulose in Pharmaceutical Industry is multifaceted, providing essential support in drug formulation and delivery.
MCC is used in tablets because it provides excellent compressibility, binding strength, and disintegration performance. Therefore,It allows manufacturers to produce strong tablets while maintaining rapid drug release.
MCC functions as both a binder and a disintegrant. During compression, it binds particles together to form a strong tablet. After administration, it absorbs water and promotes tablet disintegration.
The main advantages of MCC include superior compressibility, good flowability, rapid disintegration, compatibility with many APIs, support for direct compression manufacturing, and consistent pharmaceutical performance.
We support bulk supply, COA, DMF documentation, and formulation technical support.
Response time: within 24 hours
MOQ: 25 kg – 500 kg samples available
Global export: EU / US / Asia / LATAM