

Is MCC safe?
✔ Yes, it is considered safe by FDA, EFSA, and USP
✔ It is not absorbed by the human body
✔ It functions as dietary fiber and excipient
Yes — microcrystalline cellulose safety is well established by global regulatory authorities including FDA GRAS status and EU E460 approval.
If you have ever checked the ingredient label on a tablet, supplement, or processed food, you have likely seen microcrystalline cellulose (MCC). Despite its widespread use, many people still ask: is microcrystalline cellulose safe?
This article provides a clear, evidence-based explanation of microcrystalline cellulose safety, including FDA GRAS status, E460 regulation, digestion behavior, and potential side effects.
If you have ever checked the ingredient label on a tablet, a low-fat salad dressing, or a powdered supplement, there is a strong chance you have encountered microcrystalline cellulose (MCC). It appears in hundreds of everyday products, yet many consumers and even formulation professionals find themselves asking the same question: is microcrystalline cellulose actually safe to consume?
The answer is yes — and it is not a matter of opinion. Microcrystalline cellulose has been evaluated and approved.
It is also continuously monitored by regulatory authorities.It carries FDA GRAS (Generally Recognized As Safe) status, is listed as food additive E460 under European Union regulations, and is included in the United States Pharmacopeia (USP), European Pharmacopoeia (EP), and Food Chemicals Codex (FCC). No credible regulatory body worldwide has identified microcrystalline cellulose as a health risk at normal consumption levels.
This article provides a complete, evidence-based breakdown of microcrystalline cellulose safety — covering its regulatory status, how the body processes it, potential side effects at extreme doses, and its established role across food and pharmaceutical applications.
Microcrystalline cellulose is a purified, plant-derived material. It is widely used as a food additive (E460) and pharmaceutical excipient.
It is produced through controlled acid hydrolysis. This process removes amorphous regions and leaves a stable crystalline structure.
Unlike synthetic additives, MCC is plant-based. It does not contain active biological compounds and does not react with APIs or nutrients.
| Regulatory Body | Status |
|---|---|
| FDA | GRAS (21 CFR 182.90) |
| EU | E460 approved |
| USP | Pharmaceutical excipient |
| EFSA | Safe for food use |
The FDA GRAS designation is not granted casually. It requires strong scientific consensus.
GRAS (Generally Recognized As Safe) means an ingredient is considered safe under intended use conditions based on scientific evidence and expert evaluation.
Microcrystalline cellulose has maintained GRAS status under 21 CFR 182.90 for decades. It is widely used as a binder, stabilizer, and texturizer in food products.
The FDA does not assign a strict ADI for MCC. This indicates that normal consumption levels present no safety concern.
n pharmaceutical applications, MCC is a key multifunctional excipient used in tablet formulation, capsule filling, and controlled-release systems.
👉 Explore pharmaceutical MCC applications, grades, and formulation guides in our pharmaceutical cellulose excipients hub
| Ingredient | Safety | Source | Function |
|---|---|---|---|
| MCC (E460) | Very High | Plant | Fiber / stabilizer |
| Silica | High | Mineral | Anti-caking |
| Starch | High | Plant | Thickener |
In the European Union, microcrystalline cellulose is classified under food additive number E460(i), with powdered cellulose designated as E460(ii). Both fall under the broader category of cellulose-based food additives regulated by the European Food Safety Authority (EFSA).
EFSA has conducted multiple safety assessments of cellulose-based additives and consistently concluded that MCC is safe for use in food at technologically justified levels. It is authorized across a wide range of food categories under Regulation (EC) No 1333/2008, including processed cheese, dietary foods for specific nutritional uses, and tablet or capsule-form food supplements.
The dual approval from both the FDA (GRAS) and EFSA (E460) provides a global regulatory consensus that very few food additives achieve. For manufacturers supplying products to international markets, this alignment simplifies compliance and reinforces the safety credentials of MCC as an ingredient.
Understanding MCC safety requires understanding what the body actually does with it — and the answer is: essentially nothing, by design.
Microcrystalline cellulose is an insoluble dietary fiber. Humans do not produce the enzyme cellulase, which would be required to break down the beta-1,4-glycosidic bonds in cellulose chains. As a result, MCC passes through the gastrointestinal tract largely intact, is not absorbed into the bloodstream, and is excreted without systemic exposure.
This non-absorbable nature is not a deficiency — it is the property that makes MCC both safe and functionally useful:
Long-term animal studies conducted at doses far exceeding any realistic human exposure have found no evidence of carcinogenicity, mutagenicity, reproductive toxicity, or organ damage attributable to MCC. These studies form part of the scientific foundation underlying the global regulatory consensus on its safety.
Colloidal microcrystalline cellulose — sometimes labeled as CMCC or MCC/CMC — is a hydrated, dispersible form of MCC co-processed with a small amount of carboxymethylcellulose sodium. It is used primarily as a colloidal MCC suspension stabilizer and thickening agent in liquid food systems, pharmaceutical suspensions, and cosmetic gels.
The safety profile of colloidal MCC is consistent with standard MCC. The hydration and dispersion process does not chemically alter the cellulose backbone or introduce new reactive species. EFSA and the FDA evaluate the colloidal form under the same regulatory framework as standard MCC, and both components — MCC and CMC — carry independent safety approvals.
From a formulation standpoint, colloidal MCC functions as a food stabilizer that prevents sedimentation, maintains homogeneity, and delivers a smooth texture in products such as chocolate milk, sauces, ice cream, and liquid nutritional supplements. Its safety has been validated both as a standalone ingredient and in combination with other approved food additives.
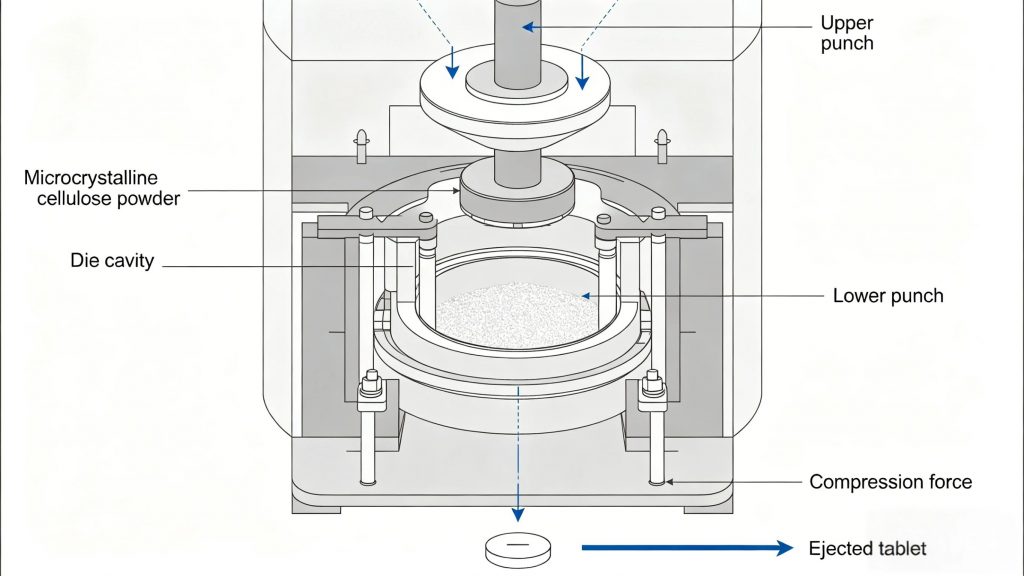
The pharmaceutical industry’s adoption of MCC as a primary excipient is itself a powerful safety signal. Pharmaceutical-grade MCC must meet the stringent purity and quality standards set by the USP/NF, EP, and JP (Japanese Pharmacopoeia), including defined limits for heavy metals, residue on ignition, microbial contamination, and particle size distribution.
In tablet and capsule manufacturing, microcrystalline cellulose serves multiple critical functions — as a binder, a direct compression excipient, a controlled-release matrix component, a capsule filler, and a disintegration enhancer. Across all of these roles, its chemical inertness ensures that it does not interact with active pharmaceutical ingredients, alter their pharmacokinetics, or introduce degradation products that could affect patient safety.
Regulatory agencies including the FDA, EMA, and PMDA have approved MCC-containing drug products across virtually every therapeutic category. Its presence in the FDA’s Inactive Ingredient Database reflects decades of accepted use in approved drug applications at established, well-characterized concentration ranges.
For manufacturers seeking a pharmaceutical excipient that combines functional versatility with an unimpeachable safety record, MCC remains the industry benchmark.
For the vast majority of people, microcrystalline cellulose consumed through normal food or pharmaceutical products produces no side effects whatsoever. This is consistent with its regulatory status and its non-absorbable nature.
However, at unusually high intake levels — well beyond what would be encountered through normal consumption — some individuals may experience:
These effects are not specific to MCC and are characteristic of any significant increase in insoluble fiber intake. They are transient, dose-dependent, and have not been associated with structural or functional harm to the gastrointestinal tract.
No credible clinical evidence links MCC to allergic reactions, immune responses, endocrine disruption, or systemic toxicity under normal or elevated dietary conditions. The rare case reports of digestive sensitivity in the literature involve doses that are not achievable through normal consumption of food or pharmaceutical products.
Microcrystalline cellulose functions as a food stabilizer and thickening agent cellulose in a broad spectrum of processed food categories. Common applications include:

MCC helps prevent whey separation and improves texture in yogurt and milk drinks.
It improves powder flow and extends shelf life in baking ingredients.
MCC is used as a tablet binder and capsule filler in nutraceutical products.
In sauces and ready meals, MCC stabilizes texture and prevents ingredient separation.
Yes. Microcrystalline cellulose is considered safe by global regulatory authorities.
The FDA classifies it as GRAS under 21 CFR 182.90. This means it is safe under intended use conditions.
This conclusion is based on decades of scientific research and regulatory review.
Microcrystalline cellulose safety is not uncertain. It has been clearly established worldwide.
Microcrystalline cellulose is derived from purified plant cellulose, typically sourced from wood pulp or cotton linters. It is not synthetic and does not originate from petrochemical processes.
No. MCC is an insoluble dietary fiber. Humans lack the enzyme necessary to break down cellulose, so MCC passes through the digestive tract without being absorbed into the bloodstream.
Yes. Colloidal microcrystalline cellulose is a hydrated dispersion co-processed with carboxymethylcellulose sodium, designed for use in liquid systems, gels, and suspension formulations. Standard MCC is a dry powder used primarily in tablets, capsules, and dry food applications.
Does Microcrystalline Cellulose Have Side Effects?
Microcrystalline cellulose is well tolerated at normal intake levels.
It does not cause side effects in typical food or pharmaceutical use.
There is no evidence linking MCC to toxicity, allergies, or organ damage.
At very high intake levels, some individuals may experience mild symptoms.
These include:
These effects are common for all insoluble fibers.
They are mild, temporary, and dose-dependent.
They do not cause long-term harm.
Microcrystalline cellulose safety in food applications is supported by both its regulatory status and its fundamental biological behavior. Listed as food additive E460(i) under European Union Regulation (EC) No 1333/2008 and approved by EFSA following multiple independent safety assessments, MCC is authorized for use across a wide range of food categories — including dairy products, baked goods, processed foods, dietary supplements, and low-calorie formulations.
The reason microcrystalline cellulose safety in food is so well established comes down to a single biological fact: humans cannot digest it. MCC is an insoluble dietary fiber with no caloric value. the human body does not produce cellulase — the enzyme required to break down cellulose bonds — MCC passes through the gastrointestinal tract entirely intact, is never absorbed into the bloodstream, and exits the body without systemic exposure. It does not interact with nutrients, does not interfere with vitamin or mineral absorption, and does not undergo metabolic transformation into any biologically active compound. This complete physiological inertness is precisely why microcrystalline cellulose safety in food has never been a point of serious scientific contention — the ingredient simply does not engage with the body in any way that could produce harm under normal consumption conditions.
As a professional microcrystalline cellulose manufacturer and bulk supplier, we provide pharmaceutical grade MCC, food grade MCC, and colloidal MCC for global industries including pharmaceuticals, food processing, and nutraceuticals.
MCC is part of a broader cellulose derivatives family widely used in food and pharmaceutical industries.
👉 View cellulose derivatives including HPMC, CMC, HEMC, and modified starch systems for formulation optimization
Microcrystalline cellulose occupies a rare position in the world of food and pharmaceutical ingredients: it is simultaneously one of the most extensively used and one of the most thoroughly validated materials in both industries. Its safety is not an assumption — it is the product of decades of regulatory review, independent scientific research, and real-world use across billions of consumer products globally.
From FDA GRAS certification to E460 authorization, from USP compliance to EFSA assessment, the global regulatory consensus on MCC is consistent and unambiguous. For consumers, it is a safe, inert, plant-derived ingredient. For formulators and manufacturers, it is the pharmaceutical excipient and food stabilizer against which alternatives are measured.
Whether your interest is consumer safety, formulation science, or regulatory compliance, the evidence on microcrystalline cellulose points in one direction: it is among the safest and most well-characterized ingredients in the modern food and pharmaceutical supply chain.