Colloidal microcrystalline cellulose (MCC gel) is a suspension stabilizer that forms a three-dimensional gel network to prevent particle sedimentation in liquid systems.Colloidal MCC stabilizer, also known as MCC gel E460i, is a highly functional microcrystalline cellulose E460i system widely used as an E460(i) stabilizer in beverage, dairy, and pharmaceutical formulations. Compared with traditional hydrocolloids, MCC gel provides superior suspension stability and texture control.
Related solutions: colloidal MCC stabilizer | microcrystalline cellulose excipient

Colloidal microcrystalline cellulose (E460(i) stabilizer) is a structured cellulose-based suspension system engineered through making from microcrystalline cellulose and carboxymethyl cellulose (CMC). It functions as a three-dimensional thixotropic network system for stabilizing insoluble particles in liquid formulations.
This material is widely recognized as a functional suspension and stabilization system in food, pharmaceutical, and industrial applications. Unlike conventional thickeners that only increase viscosity, colloidal MCC builds a physical gel architecture that holds particles in permanent suspension — regardless of processing stress, heat, or storage conditions.
Why Leading Manufacturers Choose Colloidal MCC
– Proven suspension stability across food and pharma systems
– Cleaner label alternative to synthetic stabilizers
– Reliable performance under heat, shear, and low pH
– Direct manufacturer supply with consistent quality
Colloidal MCC is considered one of the most effective cellulose-based suspension systems in modern formulation science.
👉 Request technical data sheet or sample for formulation testing
Colloidal MCC was developed to solve a fundamental formulation challenge shared across food science, pharmaceutical development, and industrial engineering:
How do you keep insoluble particles stable inside a liquid system — permanently — without sedimentation, phase separation, or texture collapse over time?
Traditional stabilizers such as starch, CMC alone, or HPMC address viscosity but cannot build a true structural network. Colloidal MCC solves the problem at the architectural level: it creates a three-dimensional gel framework that physically holds particles in suspension.
This is achieved through three interdependent mechanisms:
1. Particle Network Entanglement Microfibrils interlock to form a continuous load-bearing matrix throughout the liquid system.
2. Water Immobilization Structure Free water is bound within the network, eliminating the sedimentation pathways that cause particle settling.
3. Shear-Thinning Gel Behavior The system flows easily under mechanical stress and rebuilds its structure at rest — the defining property known as thixotropy.
| TEST ITEMS | ACT591 | ACT3212 | ACT611 | ACT538 | ACT521 |
| Loss on drying,w/% | ≤7.0 | ≤7.0 | ≤7.0 | ≤7.0 | ≤7.0 |
| Residue on ignition,w/% | ≤5.0 | ≤5.0 | ≤5.0 | ≤5.0 | ≤5.0 |
| Viscosity,1.2%,mpa.s | 39-91 | 50-200 | 50-151 | 39-175 | 50-100 |
| Particle Size retained on 60 mesh sieve | <1 | <1 | <1 | <1 | <1 |
| Heavy Metal,mg/kg | ≤10 | ≤10 | ≤10 | ≤10 | ≤10 |
| Total aerobic microbial count,cfu/g | ≤1000 | ≤1000 | ≤1000 | ≤1000 | ≤1000 |
| Total moulds and yeasts count,cfu/g | ≤100 | ≤100 | ≤100 | ≤100 | ≤100 |
| Escherichia coli | Not detected/10g | Not detected/10g | Not detected/10g | Not detected/10g | Not detected/10g |
| Salmonella species | Not detected/10g | Not detected/10g | Not detected/10g | Not detected/10g | Not detected/10g |
| Staphylococcus aureus | Not detected/10g | Not detected/10g | Not detected/10g | Not detected/10g | Not detected/10g |
| Pseudomonas aeruginosa | Not detected/10g | Not detected/10g | Not detected/10g | Not detected/10g | Not detected/10g |
| Application | food and beverage | Milk shake, sauce | food and beverage | Neutral milk drinks, vegetable protein drinks |
food and beverage |
– Appearance: white to off-white powder
– Particle size: controlled microcrystalline distribution
– pH (1% solution): 6.0–8.0
– Loss on drying: ≤ 7.0%
– Recommended dosage: 0.1%–1.0% depending on application
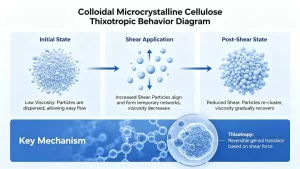
When dispersed in water, colloidal MCC undergoes a hydration and network-forming process. The CMC component hydrates first, separating MCC microfibrils and allowing them to form a stable colloidal suspension. The result is a reversible, thixotropic gel system with three distinct behavioral states:
At rest: Microfibrils re-entangle into a three-dimensional network, holding all particles in fixed, uniform suspension.
Under shear: The network disrupts and viscosity drops, allowing the system to flow freely through pumps, mixers, and homogenizers during production.
After shear: Structure rebuilds spontaneously — no external stimulus required. The gel network re-forms at rest, restoring long-term stability in the final product.
This thixotropic behavior is what distinguishes colloidal MCC from simple thickeners. It maintains suspension stability through heat sterilization, acidic pH environments, high-shear mixing, and freeze-thaw cycles — conditions that degrade starch- and gum-based systems.
E460(i) stabilizer (E460(i) stabilizer) is widely used in beverage stabilizer and ice cream stabilizer systems. Colloidal MCC stabilizer forms a three-dimensional network that prevents sedimentation and improves mouthfeel.E460(i) stabilizer (MCC gel) is widely used in beverage stabilizer and ice cream stabilizer systems.
In food manufacturing, colloidal MCC is critical in any product where insoluble particles, fats, or proteins must remain evenly distributed throughout shelf life.
Ice Cream Texture Stabilization Prevents ice crystal growth and fat phase separation during storage and temperature fluctuation. Delivers consistent texture from factory to consumer.

Plant-Based Milk Suspension Keeps insoluble oat, almond, or soy particles homogeneously distributed in low-viscosity systems. Eliminates the sedimentation layer that forms in unstabilized plant milks.
Dairy Beverage Uniformity Maintains protein and mineral suspension in UHT-processed dairy drinks throughout extended shelf life.
Sauce and Emulsion Stabilization Provides long-term emulsion stability in dressings, gravies, and blended sauces. Prevents oil-water separation and particle settling without heavy gum loading.
Cellulose-based stabilizers are widely used in suspension systems for food and pharma formulations. See general reference: FDA Food Additives Status List
In pharmaceutical liquid formulations, colloidal MCC provides the precision suspension stability required for dose accuracy and patient compliance.For pharmaceutical formulations, MCC is widely used in pharmaceutical excipient solutions.
Oral Suspension Formulation Ensures uniform API distribution in each dose unit — critical for therapeutic consistency in antibiotics, antacids, and pediatric medications.
Pediatric Liquid Stability Maintains stable, easy-to-redisperse suspensions in syrups and drops designed for children, where accurate dosing is non-negotiable.
API Dispersion Systems Prevents active ingredient sedimentation in low-solubility API liquid formulations where conventional stabilizers are insufficient.
Controlled Viscosity Liquids Delivers target viscosity profiles suitable for oral delivery or topical pharmaceutical application.
Paint and Coating Suspension Prevents pigment settling and maintains even color distribution through extended storage periods. Reduces the separation problems common in water-based paints.
Ceramic Slurry Stabilization Controls rheology and particle settling in casting slips and glazes, improving consistency in ceramic manufacturing.
Cosmetic Emulsions Stabilizes oil-in-water systems and suspends insoluble actives in lotions and serums, contributing to long shelf life and smooth texture.
Personal Care Gels Provides elegant, thixotropic texture in gels, cleansers, and leave-on products while keeping any suspended particles uniformly distributed.
Formulation scientists consistently select colloidal MCC over conventional stabilizers for four fundamental performance reasons. These are not marketing claims — they reflect structural properties that make MCC gel categorically different from viscosity-based systems.
Structure, Not Just Viscosity MCC gel creates a physical framework that holds particles in place. Conventional thickeners only increase resistance to flow — they cannot prevent settling once a product is at rest on a shelf.
Long-Term Sedimentation Prevention The three-dimensional network remains intact through the product’s full shelf life, including in high-density particle systems where other stabilizers fail progressively over time.
Processing Condition Resilience Colloidal MCC performs reliably under heat sterilization, acidic pH, high-shear mixing, and freeze-thaw cycling — the exact conditions that degrade starch- and gum-based systems most quickly.
Clean-Label Functional System As a cellulose-derived ingredient (E460ii), MCC gel meets clean-label expectations while delivering the technical performance of more complex synthetic stabilizer blends. It requires no synthetic additives or chemical modification at point of use.
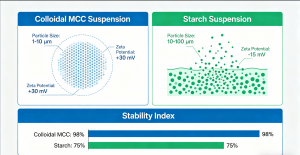
Understanding where colloidal MCC sits relative to other common stabilizers is essential for correct formulation decisions.
Colloidal MCC vs. CMC CMC is a solubility-based thickener. It increases viscosity but cannot form a structural suspension network. Colloidal MCC uses CMC as a structural component within a co-processed architecture — the two materials function at entirely different levels. CMC thickens; colloidal MCC builds.
Compared with CMC, MCC gel provides a true suspension network rather than simple viscosity. As an E460(i) stabilizer, colloidal MCC stabilizer delivers better long-term stability.Compared with CMC, MCC gel provides better suspension stability. Learn more from our CMC supplier page.
Colloidal MCC vs. Starch Starch provides short-term, swelling-based viscosity that breaks down under heat, shear, and acidic conditions. It offers no long-term suspension architecture. Colloidal MCC is stable under all three of these conditions and rebuilds its network after mechanical disturbance.
Colloidal MCC vs. HPMC HPMC is a viscosity modifier and film former, useful in solid dosage forms and coatings. In liquid systems, it provides viscosity-dependent suspension only — as viscosity decreases over time, suspension performance declines in parallel. Colloidal MCC maintains suspension through structural entanglement rather than viscosity alone.
Colloidal MCC vs. Xanthan Gum Xanthan gum offers a partial gel network, but it is sensitive to salt concentration, high-acid environments, and heat cycling. Colloidal MCC provides more consistent long-term performance across a wider range of processing conditions.
Key Distinction: Colloidal MCC is a suspension architecture system. All other listed stabilizers are primarily viscosity management tools — they differ from MCC gel in mechanism, not merely degree of effectiveness.
Compared to HPMC, colloidal MCC provides structural suspension rather than viscosity-only modification.
Colloidal microcrystalline cellulose is recognized across food technology, pharmaceutical formulation science, and industrial process engineering as the reference-class multi-functional suspension system for replacing unstable or single-function stabilizers in liquid product development.
Its designation as E460(ii) in the EU food additive framework reflects regulatory acceptance as a safe, functional, and technologically justified ingredient. In pharmaceutical applications, colloidal MCC is listed in major pharmacopeias including the USP, EP, and JP, confirming its role as a global standard excipient for liquid formulations.
Colloidal MCC (MCC gel) is used primarily to stabilize insoluble particles in liquid formulations across food, pharmaceutical, and industrial sectors. Its applications include oral suspension stabilization in medicines, texture control in dairy and plant-based beverages, emulsion stabilization in sauces, and rheology management in cosmetics and coatings. The defining characteristic is its ability to prevent sedimentation and phase separation not through viscosity alone, but through formation of a structural gel network.For liquid systems, it is recommended to use colloidal MCC stabilizer instead of standard MCC powder.
Yes. Colloidal MCC is classified as E460(ii) in the European Union’s food additive framework and is approved for use in food by regulatory authorities in the US (FDA GRAS), EU, and many other jurisdictions. In pharmaceutical applications, it is listed in the USP, EP, and JP pharmacopeias. It is derived from plant cellulose and passes through the digestive system without absorption or accumulation.For regulatory reference, colloidal microcrystalline cellulose is classified as E460(ii) in the EU food additive system. More details can be found at the EU Food Additives Database.
Microcrystalline cellulose (MCC) in powder form is used as a dry binder, filler, or disintegrant in solid dosage forms such as tablets. It does not form a functional suspension system when hydrated. Colloidal MCC (MCC gel) is a co-processed product combining MCC with carboxymethyl cellulose (CMC). This co-processing enables the formation of a thixotropic gel system specifically designed for liquid formulations. They share a base material but serve entirely different formulation functions.
Carboxymethyl cellulose (CMC) serves as the dispersing and hydration agent in colloidal MCC. Without CMC, MCC microfibrils cannot separate adequately in water — they would aggregate and settle instead of forming a network. CMC coats the MCC microfibrils during the co-processing stage, enabling them to hydrate individually and form the interlocked three-dimensional network responsible for suspension stability. CMC does not function as a standalone thickener in this system; it is structurally integrated into the MCC gel architecture.
Thixotropy is the property of a gel to become less viscous when mechanically disturbed — during pumping, mixing, or shaking — and to recover its gel structure when the disturbance stops. For colloidal MCC, thixotropy is the defining functional property: it allows the suspension system to survive manufacturing processes (high-shear mixing, homogenization, pumping) without permanent structure loss, then rebuild the gel network in the final product container — maintaining long-term particle suspension throughout shelf life.
Usage levels vary by application. In beverages and dairy systems, typical inclusion levels range from 0.1% to 0.5%. In sauces and emulsions, levels of 0.3% to 1.0% are common. Pharmaceutical oral suspensions may use higher levels depending on API characteristics. Always consult your supplier’s technical data sheet for application-specific dosage guidance.
Yes. MCC gel provides long-term structural stability, while starch only provides temporary viscosity.
Yes. It maintains stability under low pH conditions where many stabilizers fail.
Food, pharmaceutical, cosmetics, and coatings industries widely use it.
Usually between 0.1% and 1.0% depending on formulation requirements.
MCC gel E460i is a colloidal MCC stabilizer system made from microcrystalline cellulose E460i and CMC.

Colloidal MCC (E460ii) · Functional Suspension System · Food · Pharmaceutical · Industrial
Reviews
There are no reviews yet.