
CMC uses in pharmaceuticals are essential for tablet binding, suspension stabilization, and controlled drug delivery systems
PCarboxymethyl cellulose (CMC), also known as E466, is a water‑soluble cellulose derivative. Pharmaceutical formulators use it in tablets, syrups, ophthalmic solutions, and topical formulations. They employ CMC as a binder, suspension stabilizer, thickener, controlled‑release matrix former, and ophthalmic lubricant. It improves tablet strength, stabilizes liquid formulations, and controls drug release profiles.
Carboxymethyl cellulose (CMC), also known as E466, is a water‑soluble excipient. Its safety, stability, and versatility lead formulators to use it widely in tablets, syrups, ophthalmic solutions, and topical formulations.
In this guide, you’ll learn:
✔ 7 key uses of CMC in pharmaceutical formulations
✔ How CMC compares with MCC and HPMC
✔ How to choose the right viscosity grade
According to FDA GRAS database and pharmacopeia standards, CMC is widely approved for pharmaceutical use.
Explore our CMC supplier solutions for pharmaceutical applications.
Carboxymethyl cellulose (CMC), also known as E466, is a water‑soluble cellulose derivative. Formulators widely use it across oral, topical, and ophthalmic drug formulations.
CMC exhibits safety and non‑toxicity, and the body does not systemically absorb it. This characteristic makes it suitable for oral, topical, and ophthalmic applications. All major pharmacopeias including USP‑NF, EP, JP, and BP list it, and the FDA grants it GRAS status.. CMC in pharmaceuticals possesses a combination of water solubility, viscosity‑building capacity, pH stability, and broad compatibility with other pharmaceutical ingredients. This combination makes it one of the most versatile excipients available across multiple dosage forms.
Understanding how CMC works is essential for formulators. So is understanding how to select the right grade for applications across tablets, syrups, ophthalmic solutions, controlled-release systems, and topical products..
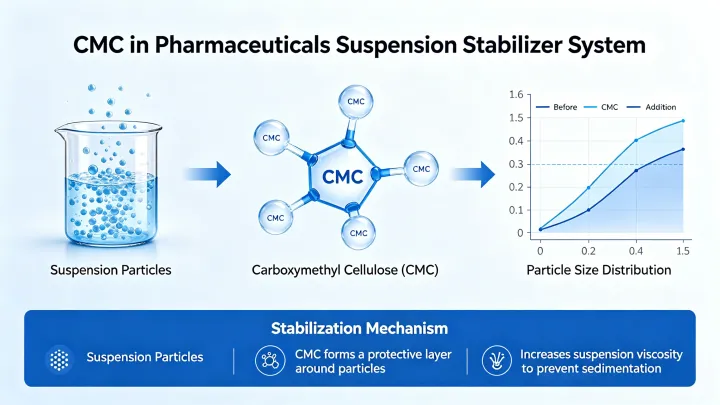
Pharmaceutical formulators widely use carboxymethyl cellulose (CMC) in suspensions. Specifically, they do so because CMC provides stable viscosity control and improves uniformity of dispersed drug particles.
Furthermore, pharmaceutical suspensions require consistent dosing accuracy, since active pharmaceutical ingredients (APIs) tend to settle under gravity over time. Therefore, without a stabilizing agent such as CMC, dose uniformity suffers. As a consequence, the first and last doses from a bottle may contain significantly different API concentrations.
In addition, CMC improves suspension stability by increasing the resistance of particles to sedimentation. At the same time, it maintains a pourable viscosity suitable for oral administration.
As a result, formulators prefer CMC in pediatric syrups, antibiotic suspensions, and antacids. Additionally, they also prefer it in other liquid dosage forms where dose uniformity is critical.
CMC controls viscosity through polymer chain hydration and entanglement in aqueous systems.
Initially, when dispersed in water, CMC molecules hydrate and expand, forming a three-dimensional network that increases the viscosity of the liquid phase. Furthermore, this network slows down particle movement, reducing sedimentation rate.
Next, in addition, CMC exhibits pseudoplastic (shear-thinning) behavior, meaning viscosity decreases during shaking or pouring but recovers at rest. Therefore, the suspension remains easy to administer while maintaining long-term stability.
Consequently, as a result, CMC provides a balance between high storage stability and good patient usability, which is essential in pharmaceutical liquid formulations.
Finally, moreover, this viscosity behavior ensures uniform redispersion after shaking, which improves dosing consistency across the entire product lifecycle.
Pharmaceutical manufacturers widely use CMC as a binder, stabilizer, thickener, suspension stabilizer, and controlled-release excipient across multiple dosage forms. Each application places different demands on CMC grade, viscosity, and concentration — and selecting the wrong grade is one of the most common formulation errors in pharmaceutical development.
Tablet manufacturers use CMC as a binder in tablet manufacturing, especially in wet granulation and direct compression processes.
It helps form strong bonds between particles, improving tablet hardness, friability, and physical stability throughout shelf life.
In wet granulation, CMC is dissolved in water and used as the granulating liquid. As the granules dry, CMC forms solid bridges between particles that hold the granule structure together. The resulting granules have good flowability and compressibility, producing tablets with consistent weight and content uniformity at the tablet press.
CMC is particularly effective for poorly compressible APIs, ensuring consistent tablet quality and performance where the active ingredient itself contributes little binding strength. Therefore, the viscosity grade and degree of substitution of CMC directly influence granule strength, tablet hardness, and disintegration time — parameters that formulators must evaluate systematically during formulation development.
Pharmaceutical formulators widely use CMC as a suspension stabilizer. They use CMC to stabilize pharmaceutical suspensions by increasing liquid viscosity and preventing particle sedimentation. Without a stabilizer, insoluble drug particles settle rapidly in aqueous suspension, leading to dose inaccuracy and potential patient safety concerns.
CMC stabilizes suspensions by increasing the viscosity of the continuous aqueous phase, reducing the rate of particle settling according to Stokes’ law. However, higher CMC concentration and higher viscosity grade provide greater suspension stability — but formulators must balance this against pourable viscosity and ease of redispersion before use.
Manufacturers commonly use CMC in oral suspensions, pediatric formulations, and topical liquid systems where uniform dosing is critical. Furthermore, for pediatric patients in particular, suspension accuracy directly links to therapeutic safety, making the choice of CMC grade a clinically relevant formulation decision.
Specifically, CMC functions as a thickener in oral liquids such as syrups, elixirs, and solutions. It improves viscosity, enhances mouthfeel, and supports accurate dose measurement — three factors that directly affect patient compliance and product quality.
Moreover, CMC provides stable viscosity across a broad pH range (approximately 4–10) and does not degrade during standard liquid pharmaceutical manufacturing processes. It is compatible with common pharmaceutical sweeteners, flavors, preservatives, and active ingredients used in syrup formulations.
For pediatric and geriatric formulations where swallowing difficulty is a concern, formulators can precisely adjust CMC thickening to improve swallowability without compromising dose accuracy or product stability.
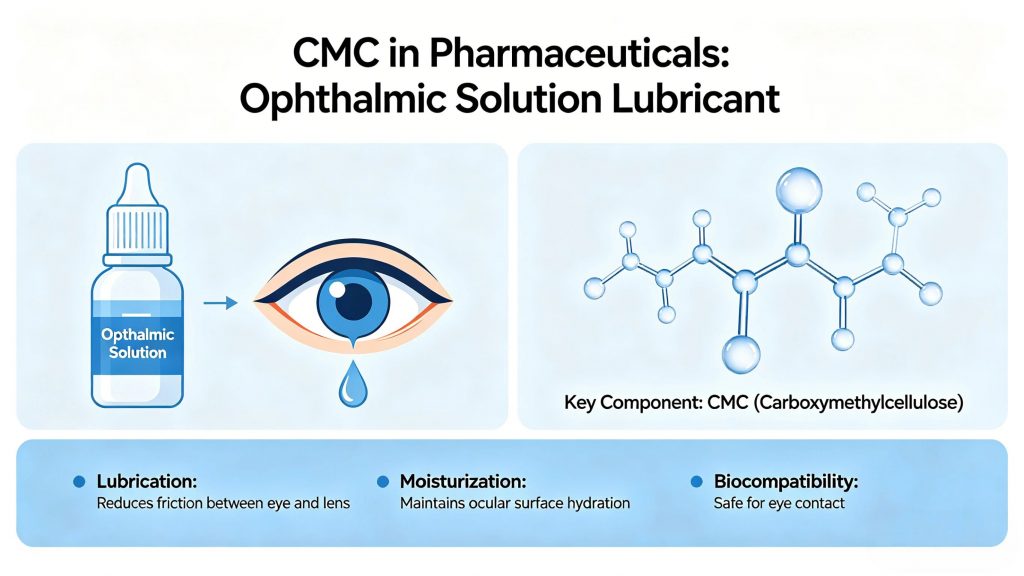
Pharmaceutical manufacturers commonly use CMC in ophthalmic solutions.
Generally,Ophthalmic preparations most widely employ CMC as a viscosity agent, including artificial tears, lubricating eye drops, and ophthalmic drug delivery systems.
Alternatively, for the second sentence: Formulators most widely use CMC as a viscosity agent in ophthalmic preparations, including artificial tears, lubricating eye drops, and ophthalmic drug delivery systems.Its unique combination of water retention, mucomimetic properties, ocular biocompatibility, and tolerability makes it the standard ingredient in this category.
In artificial tear formulations, CMC increases the residence time of the solution on the ocular surface by increasing tear film viscosity and slowing drainage rate. Concentrations of 0.5% to 1.0% CMC are standard in commercial artificial tear products — providing meaningful lubrication and moisture retention without causing blurred vision or ocular discomfort.
CMC improves moisture retention and increases the residence time of eye drops, making it highly effective in dry eye treatments. Ophthalmic-grade CMC must meet strict pharmacopeial requirements for sterility, pH (6.0–8.0), osmolality, particulate matter, and endotoxin levels — requirements that standard pharmaceutical grades do not always satisfy.
Formulators use CMC in controlled-release tablets as a matrix former. For instance, upon contact with gastrointestinal fluid, the CMC forms a gel layer around the tablet core — and drug molecules must diffuse through this gel layer before the tablet releases them.
The thickness and integrity of this gel layer directly control the rate and duration of drug release.
By adjusting CMC grade and concentration, formulators can control drug release rates over periods ranging from a few hours to 12 hours or more. Formulators use CMC-based controlled-release matrices for APIs that require sustained plasma concentrations to improve therapeutic efficacy, reduce side effects, or decrease dosing frequency.
Formulators frequently combine CMC with HPMC in controlled-release matrix formulations, with each polymer contributing different release characteristics that together produce precisely tuned release profiles that meet clinical requirements.
For sustained release systems, check our HPMC supplier.
CMC is used in gels, creams, ointments, and wound dressings as a thickener, stabilizer, and film former. It helps improve texture, consistency, spreadability, and drug delivery performance in topical and semi-solid pharmaceutical formulations.
CMC gels provide a hydrophilic matrix that maintains drug contact with the skin surface and controls the rate of drug permeation into the tissue. In wound care, CMC-based hydrogel dressings absorb wound exudate, maintain a moist wound healing environment, and support autolytic debridement — properties that directly affect healing outcomes.
CMC is also used in mucoadhesive formulations for buccal, nasal, and vaginal drug delivery. Its anionic character promotes adhesion to mucosal surfaces through electrostatic and hydrogen bonding interactions, extending the residence time of the drug at the absorption site and improving bioavailability.
CMC stabilizes pharmaceutical emulsions by increasing the viscosity of the continuous aqueous phase and forming a protective interfacial film around dispersed oil droplets. These two mechanisms together reduce the rate of droplet migration, coalescence, and phase separation — extending emulsion physical stability throughout shelf life.
It is widely used in pharmaceutical creams, lotions, and oral emulsions. First, in topical emulsions, CMC also contributes positively to the texture and skin feel of the finished product. Furthermore, its broad compatibility with emulsifying agents, active ingredients, and preservatives across a wide pH range makes it a practical and flexible choice for emulsion formulation at both development and commercial scale.
Notably, these three cellulose‑based excipients are frequently compared and occasionally confused in pharmaceutical formulation.Their mechanisms, solubility profiles, and optimal applications are fundamentally different — and understanding those differences is essential for correct excipient selection.
CMC (E466) is water-soluble and anionic. It builds viscosity by dissolving and forming polymer networks in solution. It is the preferred choice for suspension stabilization, ophthalmic solutions, syrup thickening, mucoadhesive delivery, and controlled-release matrices where ionic character is advantageous.
MCC (E460i) is water-insoluble. Rather than dissolving, it forms a physical particle network in water with measurable yield stress. Furthermore, it is the global standard excipient for direct compression tablet manufacturing and simultaneously functions as a disintegrant upon tablet contact with gastrointestinal fluid. In food applications, MCC provides structural suspension stability and fat replacement.
HPMC (E464) is water-soluble and thermally gelling — it dissolves in cold water and forms a gel upon heating, the opposite of most polymers. It is the standard excipient for pharmaceutical film coating, sustained-release hydrophilic matrix tablets, and vegetarian capsule shells. In food, it is used for thermal gelation in gluten-free bakery and oil reduction in fried foods.
These excipients are complementary rather than interchangeable.Many advanced formulations use CMC and MCC together for suspension stability. Others use CMC and HPMC together for controlled-release matrix systems with precisely tuned drug release profiles.For tablet formulations, you may also explore our microcrystalline cellulose excipient.
CMC grade selection is determined by three primary parameters: viscosity, degree of substitution (DS), and purity level. Getting this selection right at the development stage prevents costly reformulation later.
Viscosity grade is the most critical parameter:
Degree of substitution (DS) affects solubility, viscosity stability at low pH, and compatibility with other formulation components. A DS of 0.65–0.95 is standard for most pharmaceutical applications. Higher DS values produce better solubility and more stable solutions under acidic conditions — relevant for formulations exposed to gastric fluid.
Purity grade must match the application requirements. Ophthalmic and parenteral applications require ultra-pure CMC with controlled endotoxin levels, strict heavy metal limits, and validated microbiological quality. Standard pharmaceutical grades meeting USP-NF or EP specifications are appropriate for oral and topical applications.
Practical selection guide:
What is CMC uses in pharmaceuticals?
CMC (carboxymethyl cellulose) is a water‑soluble cellulose derivative. Formulators use it in pharmaceutical formulations as a binder, thickener, suspension stabilizer, controlled‑release matrix former, and ophthalmic lubricant. USP‑NF, EP, JP, and BP list it, and the FDA grants it GRAS status.
Is CMC safe for pharmaceutical use? Yes. CMC is non-toxic, non-irritating, and not systemically absorbed. It has a long and well-documented history of safe use in oral, topical, and ophthalmic pharmaceutical products globally.
What is the difference between CMC and HPMC? CMC is anionic and water-soluble at all temperatures, preferred for suspension stabilization, ophthalmic applications, and mucoadhesive drug delivery. HPMC is non-ionic and thermally gelling, preferred for film coating, sustained-release matrix tablets, and vegetarian capsule manufacturing.
Formulators use low viscosity grades at 0.5%–1.0% concentration as standard for ophthalmic eye drop formulations. All ophthalmic applications require ultra-pure grades with controlled endotoxin levels.
Yes. High viscosity CMC grades form hydrophilic gel matrices that regulate drug release over extended time periods. Formulators frequently combine CMC with HPMC in controlled-release matrix systems to achieve target release profiles matched to clinical requirements.
Formulators typically set CMC concentration in oral pharmaceutical suspensions in the range of 0.5% to 2.0% by weight. This concentration depends on the required viscosity, particle load, and target redispersibility of the suspension.
👉 Source pharmaceutical-grade CMC with full USP/Ph. Eur. compliance, DMF support, and technical formulation guidance:
View product specifications and request samples →
Authority References:
Related Reading: