
Colloidal MCC stabilizer uses are essential in modern food and pharmaceutical formulations, especially as a suspension stabilizer for beverages and liquid systems. Colloidal MCC stabilizer forms a three-dimensional gel network that prevents sedimentation, improves texture, and stabilizes emulsions in dairy products, sauces, and oral suspensions.
For detailed specifications and formulation support, explore our colloidal MCC stabilizer supplier solutions.
Colloidal MCC stabilizer is a co-processed cellulose system that combines two complementary ingredients — microcrystalline cellulose (MCC) and sodium carboxymethylcellulose (CMC) — into a single functional ingredient engineered for liquid and semi-solid applications.This system is commonly categorized under cellulose-based stabilizers used in food applications.
The term “colloidal” is critical to understanding what makes this ingredient different. In its dry state, it looks like a simple white powder. But when introduced to water under shear, it transforms into a stable colloidal dispersion — a three-dimensional network of sub-micron cellulose microfibrils suspended throughout the aqueous phase. This structural network is what gives colloidal MCC its stabilizing power.
Three core functions define this cellulose gel stabilizer:
Unlike thickeners such as CMC or starch, which simply increase viscosity to slow down sedimentation, colloidal MCC stabilizer creates a physical structure that holds particles in place — a fundamentally more reliable and efficient mechanism.
This structural approach allows formulators to achieve suspension stability at much lower concentrations, reducing costs and minimizing impact on final product texture and flavor.
👉 For full technical specifications and supply options, visit our Colloidal Microcrystalline Cellulose Product Page.
Understanding the mechanism of colloidal MCC stabilizer at a molecular level is what separates expert formulators from those who struggle with inconsistent results.
In its dry form, colloidal MCC consists of MCC microfibrils coated with a thin layer of CMC. The CMC was applied during a wet co-processing step at the manufacturing stage, then the combined material was spray-dried into a stable powder. During storage, the CMC coating keeps the microfibrils from permanently bonding to each other — this is what enables efficient redispersion later.
When colloidal MCC powder is added to water and subjected to shear energy — from a high-speed mixer, rotor-stator homogenizer, or similar equipment — the following sequence occurs:
The network that colloidal MCC forms is thixotropic — it breaks down under applied shear (such as mixing, pumping, or pouring) and rebuilds at rest. This is precisely the behavior needed in food and pharmaceutical suspension systems:
This dual behavior is why colloidal MCC stabilizer is the preferred food suspension stability additive in categories ranging from chocolate milk to pharmaceutical oral suspensions.
Several factors can impair the gel network of a colloidal MCC stabilizer:
| Disruptive Factor | Effect | Mitigation |
|---|---|---|
| Insufficient shear during dispersion | Incomplete fibril separation; weak network | Use high-shear mixer; extend mix time to 8–12 min |
| High calcium/magnesium ion concentration | Ionic interference with hydrogen bonding | Pre-disperse in deionized water; add minerals after network formation |
| pH below 3.5 | CMC precipitation; network weakens | Pre-disperse before acidification; validate at final pH |
| High soluble sugar concentration (>20%) | Competes for water; reduces network strength | Use colloidal MCC at upper end of dosage range |
| Addition after emulsification | Network cannot fully form | Always add colloidal MCC to the water phase first |
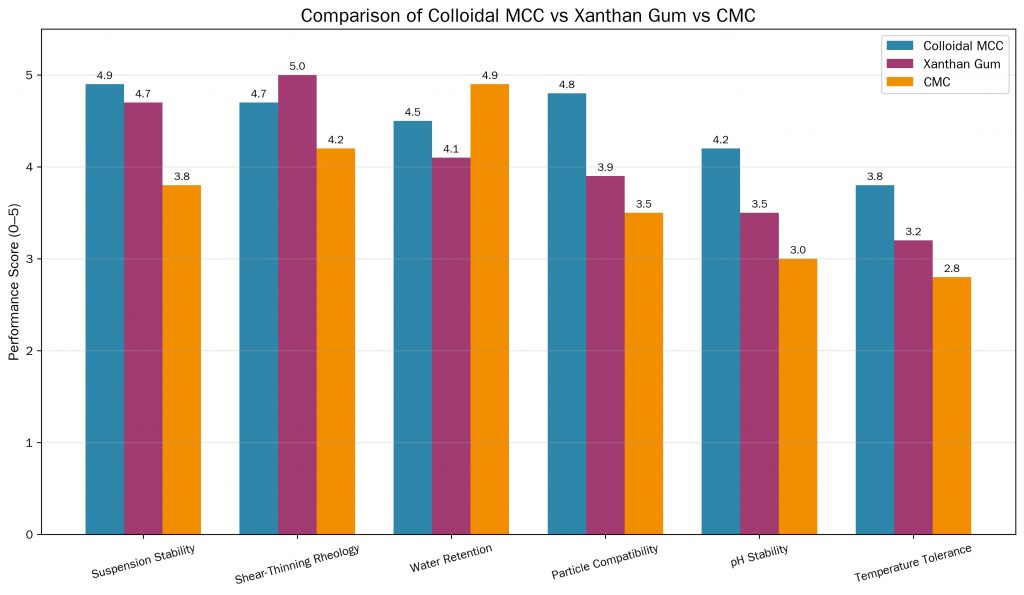
Colloidal MCC stabilizer is rarely the only hydrocolloid in a formulation. Understanding how it compares — and synergizes — with competing stabilizers is essential for optimizing both performance and cost.
Compared with carboxymethyl cellulose (CMC), colloidal MCC provides structural suspension rather than just viscosity.
| Property | CMC Alone | Colloidal MCC Stabilizer |
|---|---|---|
| Mechanism | Viscosity increase | 3D gel network |
| Suspension effect | Slows settling via viscosity | Physically traps particles |
| Dosage efficiency | Requires higher dose for stability | More effective at lower concentrations |
| Mouthfeel | Can feel slightly sticky or gummy | Smooth, creamy body |
| Shear thinning | Moderate | Strong thixotropy |
| Best use | Thickening sauces, syrups | Suspending beverages, dairy, pharma |
Synergy note: Combining colloidal MCC stabilizer (0.3–0.5%) with CMC (0.1–0.2%) often delivers superior performance compared to either ingredient alone — colloidal MCC provides structural suspension while CMC contributes additional viscosity body.
Xanthan gum is a microbially fermented polysaccharide that produces strong pseudoplastic flow behavior. It is widely used in dressings, sauces, and beverages as a suspension agent.
| Property | Xanthan Gum | Colloidal MCC Stabilizer |
|---|---|---|
| Origin | Microbial fermentation | Plant-based cellulose |
| Suspension at low dose | Very strong | Good; enhanced with co-stabilizers |
| Mouthfeel | Can feel “slimy” at higher doses | Smooth, creamy; no sliminess |
| Temperature stability | Good | Very good; heat-stable |
| Freeze-thaw stability | Moderate | Excellent |
| Cost | Higher | More economical |
| Clean label perception | Generally accepted | Plant-derived; widely accepted |
In beverage and dairy formulations, colloidal MCC stabilizer is often preferred specifically for its superior mouthfeel. High xanthan gum concentrations are detectable as a stringy or slimy texture, particularly in dairy beverages — an issue that colloidal MCC does not present.
Hydroxypropyl methylcellulose (HPMC) is a cellulose ether primarily known for its film-forming capabilities in pharmaceutical tablet coatings and as a controlled-release matrix.Compared with HPMC cellulose ethers, colloidal MCC is more suitable
| Property | HPMC | Colloidal MCC Stabilizer |
|---|---|---|
| Primary function | Film formation, gel (thermal) | Suspension stability, texture |
| Liquid suspension performance | Limited | Excellent |
| Pharmaceutical tablets | Widely used | Not primary application |
| Oral suspensions | Secondary option | Preferred option |
| Food applications | Limited | Extensive |
The verdict: For suspension stability tasks — whether in food beverages or pharmaceutical oral suspensions — colloidal MCC stabilizer significantly outperforms HPMC.
👉 Explore our full range of cellulose-based food additives: Cellulose Food Additives by Acta Biotechnology
Among the most important colloidal MCC stabilizer uses, food and beverage applications represent the largest demand segment. Here is a detailed application-by-application breakdown, including typical usage levels and key formulation considerations.
In dairy applications, colloidal MCC stabilizer solves two persistent quality problems: sedimentation of insoluble particles and syneresis (whey separation).
In chocolate milk and cocoa drinks, cocoa particles have a natural tendency to settle to the bottom within hours of production. Colloidal MCC at 0.3–0.5% creates a suspension network that keeps cocoa evenly distributed through the entire shelf life — typically 180 days in UHT-processed products.
In drinking yogurt and fermented milk beverages, protein aggregates and fruit particles must remain evenly suspended. Colloidal MCC at 0.5–1.0% prevents settling and reduces the syneresis that gives low-fat yogurt drinks a thin, watery appearance.
Critical formulation note: In high-calcium dairy systems, calcium ions interfere with the hydrogen bonding in the colloidal MCC network. Always test at your final calcium concentration and consider pre-dispersing colloidal MCC in soft water before blending into the dairy base.
Typical dosage:

One of the most important colloidal MCC stabilizer uses is in beverage suspension systems.The plant-based beverage category has driven significant growth in colloidal MCC demand. Oat milk, almond milk, soy milk, and pea protein drinks all present severe sedimentation challenges that colloidal MCC stabilizer is uniquely equipped to solve.
In oat milk, beta-glucan and insoluble oat fiber particles must remain suspended while also maintaining a clean, non-slimy mouthfeel — a balance that is difficult to achieve with xanthan gum alone. Colloidal MCC at 0.2%–0.4%, combined with gellan gum at low levels (0.01%–0.03%), achieves this balance in commercially successful formulations.
In high-protein ready-to-drink shakes, protein aggregation under UHT conditions is a major challenge. Colloidal MCC functions as a suspension stabilizer for beverages and also contributes body that protein drinks often lack without added fat.
In fortified water and mineral-enriched drinks, insoluble mineral particles (iron, calcium, zinc) require suspension. Colloidal MCC at 0.2%–0.3% is an effective food suspension stability additive for these applications.
Typical dosage:
These examples highlight why colloidal MCC stabilizer uses in beverages are critical for suspension stability.
In emulsified and semi-solid systems, colloidal MCC stabilizer performs double duty: it stabilizes the emulsion and acts as a fat replacer to deliver the texture that reduced-fat sauces often lack.
In French dressing and Italian vinaigrettes, colloidal MCC maintains oil-water suspension without the artificial thickening that starch-based stabilizers produce. At 0.5%–1.0%, it provides the viscosity and body of a full-fat dressing while significantly reducing the fat content.
In cream-based sauces and gravies, colloidal MCC stabilizer prevents phase separation during storage and post-pasteurization — a common quality issue in aseptic or retorted sauce products.
pH formulation alert: In acidic dressings (pH 3.5–4.5), the CMC component of colloidal MCC may be partially suppressed by protonation. Pre-disperse colloidal MCC in the neutral aqueous phase before acidification. Always validate suspension stability at the final pH of the product.
Typical dosage:
In semi-solid systems, colloidal MCC stabilizer uses extend to emulsion stabilization and fat replacement.
In ice cream and frozen novelty products, colloidal MCC stabilizer contributes to ice crystal control, overrun stability, and freeze-thaw resistance — three properties that define the quality of any frozen dessert.
At 0.1%–0.3%, it works synergistically with locust bean gum and carrageenan to:
Typical dosage:
As a fat replacer, colloidal MCC stabilizer exploits its gel network to mimic the creaminess, body, and mouthfeel that fat provides in food formulations. Applications include:
What colloidal MCC cannot replace: Fat’s role in fat-soluble flavor delivery and Maillard browning during baking. For applications where these are critical, partial fat replacement combined with flavor optimization is the recommended strategy.
Looking for the right colloidal MCC stabilizer for beverage or dairy formulations?
Explore our product grades or request technical support.
👉 Need help selecting the right grade for your formulation?
As a professional colloidal MCC stabilizer supplier, we provide:
👉 Contact us to discuss your application
In pharmaceutical systems, colloidal MCC stabilizer uses focus on suspension stability and dosage consistency. In oral suspensions, uniform particle distribution is not a quality preference — it is a dosing safety requirement.

In pharmaceutical formulations, colloidal MCC stabilizer uses focus on oral suspension stability.An oral suspension that allows an active pharmaceutical ingredient (API) to settle unevenly means that the first dose from a bottle may contain far less API than the last dose. For pediatric antibiotics, antifungals, antacids, and antidiarrheal suspensions, this inconsistency is a serious patient safety concern.
Colloidal MCC stabilizer is the preferred MCC stabilizer in pharma oral suspension systems for the following reasons:
Consistent network density: Unlike polymer-based thickeners that can degrade under pH or temperature stress, colloidal MCC forms a physically stable network that maintains integrity across the product’s shelf life (typically 18–24 months).
Easy redispersion: When a patient shakes the bottle before use, the colloidal MCC network rapidly breaks under shear and allows the API to redistribute uniformly — then the network rebuilds at rest. This shake-to-use behavior is a critical pharmaceutical design requirement.
API compatibility: Colloidal MCC is chemically inert with most APIs. It does not react with active ingredients or excipients under normal formulation conditions.
Regulatory acceptance: Colloidal MCC is listed in the USP-NF (National Formulary) and Ph. Eur. as an accepted pharmaceutical excipient, simplifying regulatory submissions.
Typical dosage in pharmaceutical oral suspensions: 0.5%–1.5%
These properties define key colloidal MCC stabilizer uses in oral suspensions.
In topical creams, lotions, and gels, colloidal MCC stabilizer contributes to viscosity, emulsion stability, and a smooth skin feel. For formulators pursuing clean label or natural positioning in personal care, colloidal MCC provides a plant-derived alternative to synthetic thickeners.
While less common than in oral suspensions, colloidal MCC has been explored in ophthalmic suspensions and nasal spray formulations where uniform particle distribution is critical.
👉 Developing a pharmaceutical suspension?
Our pharma-grade colloidal MCC stabilizer meets USP / EP standards and is widely used in oral suspensions.
Need pharma-grade colloidal MCC stabilizer (USP/EP compliant)?
Request samples and technical documentation.
Few food and pharmaceutical ingredients have a more thoroughly documented global safety profile than colloidal MCC stabilizer.
| Regulatory Authority | Status | Reference |
|---|---|---|
| U.S. FDA | GRAS (Generally Recognized as Safe) | 21 CFR 182.90 |
| European Union EFSA | Approved food additive | E460(ii) |
| JECFA (FAO/WHO) | No ADI established (safest classification) | JECFA Monograph |
| Japan JFSL | Approved | Japanese Food Sanitation Law |
| China | Approved | GB 2760 |
| Codex Alimentarius | Approved | INS 460(ii) |
The significance of “No ADI established”: When JECFA assigns no Acceptable Daily Intake limit to an ingredient, it means that no toxicological concern was identified at any realistic intake level during testing. This is the strongest possible safety designation that JECFA applies.
Colloidal MCC stabilizer is widely recognized as safe for use in food and pharmaceutical applications.
According to the FDA,microcrystalline cellulose is classified as Generally Recognized as Safe (GRAS) under 21 CFR 182.90.
In the European Union, the EFSA lists it as an approved food additive under E460(ii), confirming its safety for consumption across a wide range of applications.
Colloidal MCC is not digested or absorbed by the gastrointestinal tract. It passes through the digestive system intact and exits the body unchanged. As a result:
On ingredient labels, colloidal MCC appears as:
The “cellulose gel” descriptor is widely accepted by consumers in clean label audits and is generally compatible with natural-positioning products. For certified organic formulations, check with the relevant certifying body, as approval status varies by jurisdiction and certification program.
The viscosity generated by colloidal MCC stabilizer is non-linear:
Always allow 30–60 minutes for the network to equilibrate after dispersion before measuring viscosity. Measurements taken immediately post-mixing will underestimate final viscosity.
| pH Range | Performance |
|---|---|
| pH 3.5–8.5 | Optimal performance across all major food applications |
| pH 3.0–3.5 | Reduced viscosity; possible CMC suppression; validate carefully |
| pH < 3.0 | Not recommended; significant network disruption |
| pH > 8.5 | Generally stable; verify for specific pharmaceutical applications |
Colloidal MCC gel networks are thermally stable in a way that distinguishes them from starch- and gelatin-based systems:
Freeze-thaw stability is a significant advantage of colloidal MCC stabilizer over starch-based alternatives. Starches retrograde and release water (syneresis) after freeze-thaw cycles, producing unacceptable texture in frozen products. Colloidal MCC’s cellulose network does not retrograde, maintaining consistent texture through multiple freeze-thaw cycles.
Colloidal MCC stabilizer is compatible with and synergistic with most hydrocolloids:
Even experienced food scientists encounter preventable failures when working with colloidal MCC stabilizer. These are the eight most frequent mistakes, with practical guidance on avoidance.
What happens: Insufficient shear energy during dispersion results in incomplete fibril separation. Undispersed MCC aggregates form visible white specks, the gel network is weak, and suspension performance is far below expectations.
Solution: Use a high-shear rotor-stator mixer or homogenizer at 3,000–5,000 RPM minimum. Mix for at least 8–12 minutes. Pilot-scale equipment should be validated against lab-scale dispersion conditions before scale-up.
What happens: Adding colloidal MCC after emulsification, acidification, or high-sugar addition prevents proper network formation. The dispersion appears functional during production but fails during shelf life stability testing.
Solution: Always disperse colloidal MCC in the water phase first, before adding fats, acids, high-sugar concentrations, or other ingredients. This is the single most important processing rule for colloidal MCC.
What happens: In calcium-fortified beverages, dairy-based products, or hard water systems, calcium and magnesium ions compete with hydrogen bonding sites on MCC microfibrils, dramatically weakening the network. Products pass stability testing with deionized water in the lab but fail with the actual mineral-rich water used in production.
Solution: Test with production-grade water and actual mineral addition levels during development. Pre-disperse colloidal MCC in soft water if possible, and validate the final formulation at maximum mineral concentration.
What happens: Formulators who observe poor suspension stability instinctively increase the colloidal MCC concentration. If the root cause is inadequate dispersion rather than insufficient dosage, increasing concentration only makes texture problems worse — producing a grainy, chalky mouthfeel and no improvement in suspension.
Solution: Diagnose the failure mode first. Check dispersion conditions before adjusting dosage. A well-dispersed 0.4% colloidal MCC will outperform a poorly dispersed 1.0%.
What happens: Viscosity readings taken immediately after mixing (before the network has fully equilibrated) significantly underestimate final product viscosity. Formulators who adjust based on these readings often end up with over-thickened products after 24–48 hours.
Solution: Allow the dispersion to rest for 30–60 minutes (or longer for high-concentration systems) before measuring viscosity. Establish a standardized measurement protocol and stick to it across all development batches.
What happens: Many formulations are developed at neutral pH and then acidified later in the process. If colloidal MCC is added to an already-acidified system, CMC suppression reduces the network strength. Alternatively, formulators validate in deionized water but not in the final acidic product matrix.
Solution: Always validate colloidal MCC performance at the final pH of the product. For acidified products, pre-disperse colloidal MCC before acidification and confirm stability through the full pH reduction step.
What happens: Different commercial grades of colloidal MCC — even those with similar specifications on paper — can differ in CMC ratio, particle size distribution, and drying conditions. A direct supplier swap without re-validation can result in unexpected viscosity changes or reduced suspension performance.
Solution: When switching suppliers, treat it as a new ingredient introduction. Request reference samples, compare key specs (CMC content, particle size, 1.2% dispersion viscosity), and run full application testing before approving the new source.
What happens: Tap water, deionized water, mineral water, and plant-based milk base all behave differently as dispersion media. Laboratory results obtained in deionized water may not predict performance in a complex, mineral-rich, protein-containing production system.
Solution: Always conduct final stability validation in the actual production matrix — including production-grade water, all co-ingredients, and actual processing conditions.
For B2B procurement teams and formulators, supplier selection directly determines product consistency, formulation reliability, and supply chain stability. These are the criteria that matter most.
When evaluating suppliers of colloidal MCC stabilizer, always request and compare the following parameters:
| Specification | What to Evaluate |
|---|---|
| CMC content (%) | Typically 8–18%; higher CMC generally improves dispersibility |
| Viscosity of 1.2% dispersion (mPa·s) | Critical for lot-to-lot consistency benchmarking |
| Particle size (D50, D90) | Affects suspension behavior and mouthfeel |
| pH of 1% dispersion | Should be 6.0–8.0 for standard food-grade material |
| Moisture content / loss on drying | Affects shelf life and flowability |
| Heavy metals | Should meet pharmacopeia limits for pharmaceutical-grade |
| Microbial limits | TPC, yeast & mold, absence of Salmonella and E. coli |
All reputable colloidal MCC suppliers should provide:
Beyond technical specifications, ask prospective suppliers:
A supplier who cannot provide technical application support is a supplier who will leave you alone when formulation problems arise — and in product development, they always do.
👉 View detailed specifications, application support resources, and bulk supply options for colloidal MCC: Colloidal Microcrystalline Cellulose Powder — Colloidal Grade
As an experienced cellulose derivatives supplier, we focus on:
We are not just a supplier — we are your formulation partner.
Colloidal MCC stabilizer is used as a suspension stabilizer, texture improver, and emulsion stabilizer in food and pharmaceutical products. It prevents sedimentation in beverages, reduces syneresis in dairy products, maintains uniform API distribution in oral pharmaceutical suspensions, and acts as a fat replacer in low-calorie food formulations.
Yes. Colloidal MCC stabilizer is one of the most extensively evaluated food ingredients globally. It holds GRAS status from the U.S. FDA (21 CFR 182.90), is approved as E460(ii) in the European Union, and carries no Acceptable Daily Intake (ADI) limit under JECFA — indicating no identified toxicological concern at any realistic consumption level. It is not absorbed, not metabolized, and passes through the digestive system as insoluble dietary fiber.Global regulatory consensus — including FDA and EFSA — confirms that colloidal MCC stabilizer has no established ADI limit.
MCC powder (such as Avicel PH grades) is used in solid pharmaceutical tablets and capsules as a binder, filler, and disintegrant. It does not disperse in water. Colloidal MCC stabilizer (such as Avicel RC grades) is co-processed with sodium CMC, allowing it to form a stable three-dimensional gel network in water. It is designed exclusively for liquid and semi-solid applications — beverages, sauces, dairy, and pharmaceutical oral suspensions.
Typical usage levels are: 0.2%–0.5% for beverages and plant-based milks; 0.5%–1.0% for dairy products and dressings; 0.5%–1.5% for pharmaceutical oral suspensions. These ranges are starting points — the optimal concentration for any specific application must always be determined through formulation trials under actual processing conditions.
Partially, yes. Colloidal MCC stabilizer effectively replicates fat’s contribution to texture, creaminess, and mouthfeel in many reduced-fat applications including dairy beverages, dressings, sauces, and frozen desserts. It cannot replicate fat’s role in fat-soluble flavor delivery, heat-mediated browning, or caloric density as a satiety signal.
Yes, but with formulation considerations. High calcium content in dairy systems can reduce the gel network strength of colloidal MCC. Formulators should pre-disperse colloidal MCC in soft water before blending into dairy bases, and validate performance at the final calcium concentration of the product.
Colloidal MCC requires high-shear mixing for adequate dispersion. A rotor-stator homogenizer, high-speed disperser, or equivalent equipment operating at 3,000–5,000 RPM is typically required. Standard paddle mixers or overhead stirrers are generally insufficient for complete fibril separation. Dispersion time should be 8–12 minutes minimum.
No. Colloidal MCC is derived from plant cellulose (wood pulp or cotton linter) and contains no gluten. It is suitable for gluten-free formulations.
Overall, colloidal MCC stabilizer uses span food, beverage, and pharmaceutical industries.Colloidal MCC stabilizer is one of the most versatile, well-validated, and cost-efficient suspension stabilizers available to food and pharmaceutical formulators today. Its unique mechanism — building a physical three-dimensional network that traps particles rather than simply slowing their descent through viscosity — gives it performance advantages that viscosity-based stabilizers cannot replicate.
For formulators, the key to unlocking that performance lies in understanding the dispersion process: using the right shear energy, adding colloidal MCC at the correct processing stage, accounting for ionic interference, and validating performance in the actual product matrix.
For procurement teams, the key lies in supplier qualification: requesting the right technical specifications, validating lot-to-lot consistency, and partnering with suppliers who can offer genuine technical support beyond simply shipping product.
Whether you are developing a plant-based milk, a pediatric antibiotic suspension, a reduced-fat salad dressing, or a fortified protein shake, colloidal MCC stabilizer offers a reliable, globally approved, and consumer-accepted solution to your most challenging formulation stability problems.
Looking for a reliable colloidal MCC stabilizer supplier? Contact us for samples and technical support.
👉 Ready to source or evaluate colloidal MCC for your formulation?
👉 Request a sample or bulk quotation for your formulation
👉 Speak with our technical team for application support
Looking for a trusted colloidal MCC stabilizer supplier for your business?
We support:
✔ Food & beverage manufacturers
✔ Pharmaceutical companies
✔ Distributors & importers
With:
👉 Send us your requirements today and get a fast quotation.