We are a professional colloidal microcrystalline cellulose supplier and manufacturer with over 15 years of production experience, serving global food, pharmaceutical, and industrial markets.
We are a professional colloidal microcrystalline cellulose supplier and manufacturer, offering bulk supply with consistent viscosity control, regulatory compliance (E460, USP), and global export support.With over 15 years of manufacturing experience, we supply high-quality colloidal MCC to food, pharmaceutical, and industrial manufacturers in more than 30 countries worldwide. Our products are known for stable viscosity performance, consistent quality, and full regulatory compliance.
✔ MOQ from 1 ton
✔ Food & pharma grade available
✔ Custom viscosity grades
✔ Fast sampling & technical support
Colloidal MCC is a thixotropic microcrystalline cellulose used as a stabilizer, suspending agent, and viscosity modifier in food, pharmaceutical, and industrial formulations.
Colloidal MCC is widely used in food systems as a pre-hydrated gel. For practical formulations, see our MCC gel for food.
Colloidal MCC is derived from microcrystalline cellulose, a purified, partially depolymerized cellulose widely used in food and pharmaceutical applications.
Looking for a reliable colloidal MCC supplier?
👉 Get a free sample & technical formulation support within 24 hours

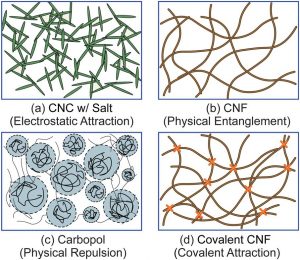
Colloidal MCC is a co-processed form of microcrystalline cellulose that has been wet-milled to sub-micron particle size and stabilized — typically with a dispersing agent such as sodium carboxymethylcellulose (CMC) — to form a thixotropic colloidal suspension when hydrated.
The term “colloidal” refers to the particle size distribution: unlike standard MCC powder, where particles are measured in tens to hundreds of microns, colloidal MCC particles are in the sub-micron to low-micron range. This dramatically increases surface area and fundamentally changes how the material behaves in liquid systems.
When dispersed in water with adequate shear, colloidal MCC particles form a three-dimensional network held together by hydrogen bonding. This network:
This combination of properties makes colloidal MCC a multi-functional stabilizer that behaves fundamentally differently from any soluble hydrocolloid.
The thixotropic behavior of colloidal MCC has been widely documented in peer-reviewed studies (ScienceDirect).
1.
| TEST ITEMS | ACT591 | ACT3212 | ACT611 | ACT538 | ACT521 |
| Loss on drying,w/% | ≤7.0 | ≤7.0 | ≤7.0 | ≤7.0 | ≤7.0 |
| Residue on ignition,w/% | ≤5.0 | ≤5.0 | ≤5.0 | ≤5.0 | ≤5.0 |
| Viscosity,1.2%,mpa.s | 39-91 | 50-200 | 50-151 | 39-175 | 50-100 |
| Particle Size retained on 60 mesh sieve | <1 | <1 | <1 | <1 | <1 |
| Heavy Metal,mg/kg | ≤10 | ≤10 | ≤10 | ≤10 | ≤10 |
| Total aerobic microbial count,cfu/g | ≤1000 | ≤1000 | ≤1000 | ≤1000 | ≤1000 |
| Total moulds and yeasts count,cfu/g | ≤100 | ≤100 | ≤100 | ≤100 | ≤100 |
| Escherichia coli | Not detected/10g | Not detected/10g | Not detected/10g | Not detected/10g | Not detected/10g |
| Salmonella species | Not detected/10g | Not detected/10g | Not detected/10g | Not detected/10g | Not detected/10g |
| Staphylococcus aureus | Not detected/10g | Not detected/10g | Not detected/10g | Not detected/10g | Not detected/10g |
| Pseudomonas aeruginosa | Not detected/10g | Not detected/10g | Not detected/10g | Not detected/10g | Not detected/10g |
| Application | food and beverage | Milk shake, sauce | food and beverage | Neutral milk drinks, vegetable protein drinks |
food and beverage |
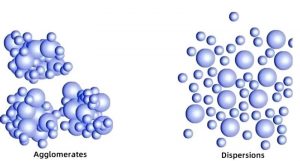
Understanding the distinction between these two forms is essential for selecting the right material for your application.
| Property | Colloidal MCC | Powder MCC |
|---|---|---|
| Particle size | Sub-micron to low-micron range | 20–200 microns (typical) |
| Physical state | Hydrated colloidal suspension (or redispersible powder) | Dry white powder |
| Activation | Requires moderate shear to activate network | No hydration needed; used dry |
| Primary mechanism | Thixotropic gel network formation | Mechanical binding, compression |
| Key function in food | Stabilizer, fat replacer, suspension agent | Dietary fiber, anti-caking |
| Key function in pharma | Suspension stabilizer, emulsion agent | Tablet binder, disintegrant |
| Regulatory status | E460(ii) – Microcrystalline cellulose (EU); GRAS (USA) | E460(i) – Microcrystalline cellulose (EU); GRAS (USA) |
| Water solubility | Insoluble, but forms stable colloidal dispersion | Insoluble; does not disperse colloidally |
| Thixotropy | Yes — shear-thinning with structural recovery | No |
The critical distinction for formulators: powder MCC contributes structure in dry or semi-dry systems (tablets, powders, dry mixes), while colloidal MCC contributes structure in liquid and semi-liquid systems (suspensions, emulsions, gels, creams) .For industrial and pharmaceutical-grade powder applications, visit our microcrystalline cellulose manufacturer page.
Compared with xanthan gum, carrageenan, or CMC, colloidal MCC offers:
• Superior suspension stability without excessive viscosity
• Clean label advantage (E460 vs multi-additive systems)
• Better freeze-thaw stability in dairy and frozen products
• Improved mouthfeel in low-fat formulations
This makes colloidal MCC the preferred choice for modern food and beverage formulations.
Colloidal MCC is used across three major industrial sectors, each exploiting a different aspect of its thixotropic, stabilizing properties.
In food manufacturing, colloidal MCC is used wherever texture, stability, and fat reduction are formulation priorities. Its E460(ii) designation covers use across a broad range of food categories under quantum satis regulations in the EU, and GRAS status permits wide application in the USA.
Ice cream and frozen desserts Colloidal MCC controls ice crystal growth during storage, stabilizes overrun (air cell structure), and improves meltdown resistance. It is particularly valuable in reduced-fat ice cream, where it compensates for the textural contribution of removed fat.Colloidal MCC is commonly used in frozen desserts. Learn more about our colloidal MCC supplier solutions for ice cream stabilization.
Low-fat and reduced-calorie dairy In yogurt, sour cream, and cheese analogs, colloidal MCC mimics fat globule behavior on the tongue, delivering lubricity and mouthfeel that make reduced-fat products perceptually comparable to their full-fat counterparts. It also suppresses surface syneresis in stirred yogurt.

Dressings, sauces, and beverages Colloidal MCC stabilizes oil-in-water emulsions in salad dressings and suspends insoluble particles in beverages such as fortified juices and plant-based milks. Its thixotropic nature ensures products flow readily when poured or shaken, then recover viscosity at rest to prevent sedimentation.In plant-based drinks, colloidal MCC ensures suspension stability. Explore our MCC gel for food solutions for plant-based milk stabilizer applications.
Bakery fillings and processed foods In custard fillings, pastry creams, and retort-processed foods, colloidal MCC maintains structural integrity during heat treatment and prevents moisture migration during storage.
Colloidal MCC holds a well-established position in pharmaceutical formulation, documented extensively in the USP/NF and Ph. Eur. pharmacopoeias. Its primary pharmaceutical functions are distinct from those of powder MCC.
Oral suspensions Colloidal MCC is the industry-standard suspending agent for oral liquid formulations containing insoluble active pharmaceutical ingredients (APIs). Its thixotropic network keeps API particles uniformly distributed throughout the suspension, preventing caking and ensuring consistent dose delivery per spoonful or measured dose.
Topical creams and gels In topical formulations, colloidal MCC stabilizes oil-in-water emulsions, contributes to skin-feel (non-greasy, smooth application), and provides the structural integrity necessary for creams to maintain consistency across storage temperatures.
Ophthalmic and parenteral suspensions In specialized pharmaceutical applications including ophthalmic drops and certain injectable suspensions, colloidal MCC’s fine particle size and colloidal stability make it suitable as a suspension medium where larger particles would be unacceptable.
Controlled-release systems Colloidal MCC is studied for use in modified-release oral dosage forms, where its network structure can modulate drug release profiles in matrix-based systems.
Beyond food and pharma, colloidal MCC finds use in industrial formulations where thixotropic behavior and particle suspension are required.
Paints and coatings Colloidal MCC is used as a thickener and anti-settling agent in water-based paints, ensuring uniform pigment distribution during storage while allowing easy application under brush or roller shear.
Personal care products In cosmetics and personal care formulations — including lotions, sunscreens, and face creams — colloidal MCC stabilizes emulsions and contributes to elegant skin feel without the use of synthetic polymers.
Agricultural formulations Colloidal MCC is used in wettable powder and suspension concentrate formulations of agrochemicals, where it keeps active ingredients suspended and improves adherence to plant surfaces after application.
Choosing the right colloidal microcrystalline cellulose (colloidal MCC) depends on your formulation goals, viscosity requirements, and processing conditions. Different grades are designed to deliver specific rheology and stability performance.
• Low viscosity grades (50–200 mPa·s)
Best for beverage systems such as milk drinks, plant-based beverages, and liquid nutritional products. These grades provide suspension stability without significantly increasing thickness, ensuring smooth drinking texture.
• Medium viscosity grades (200–800 mPa·s)
Suitable for sauces, dressings, and dairy products. They offer a balance between flowability and structure, improving mouthfeel while maintaining pourability.
• High viscosity grades (1000–8000 mPa·s)
Recommended for creams, oral suspensions, and high-solid systems. These grades create a strong three-dimensional network, delivering maximum suspension stability and thick texture.
• Beverages & plant-based milk → Focus on suspension stability and smooth mouthfeel
• Ice cream & dairy → Control ice crystals, improve creaminess, reduce fat
• Sauces & dressings → Enhance emulsion stability and viscosity control
• Pharmaceutical suspensions → Prevent sedimentation and ensure dose uniformity
• High shear processing → Select grades with fast structural recovery (strong thixotropy)
• Thermal processing (UHT / pasteurization) → Use heat-stable grades
• Low pH systems (acidic beverages) → Ensure pH-stable formulations
Selecting the optimal colloidal MCC grade often requires formulation testing. Our technical team provides:
• Viscosity matching based on your product target
• Application-specific grade recommendations
• Sample support for lab and pilot trials
• Formulation optimization for cost-performance balance
👉 Contact us to get a recommended grade for your specific application.
Q: What does “thixotropic” mean in relation to colloidal microcrystalline cellulose? A: Thixotropy describes a material’s ability to become less viscous (more fluid) under shear stress — such as shaking, stirring, or pumping — and then recover its gel-like structure when the shear force is removed. Colloidal MCC is thixotropic because its hydrogen-bonded particle network breaks apart under agitation and reassembles at rest. This is why colloidal MCC-stabilized products pour easily but do not separate or sediment during storage.
Q: How does colloidal microcrystalline cellulose differ from MCC powder? A: Colloidal MCC is a thixotropic suspension forming stable gels in aqueous systems, while powder MCC is a dry material used mainly as a tablet binder and anti-caking agent. The sub-micron particles of colloidal MCC create a hydrogen-bonded network capable of suspending particles, stabilizing emulsions, and preventing syneresis — functions that powder MCC cannot perform in liquid systems.
Q: Is colloidal MCC the same as MCC Gel? A: These terms are closely related and sometimes used interchangeably, but there is a technical distinction. “Colloidal MCC” refers to the material type — microcrystalline cellulose processed to colloidal particle size. “MCC Gel” typically refers to a specific commercial product form: a ready-to-use colloidal MCC dispersion formulated and optimized for food applications. All MCC Gel products are colloidal MCC, but not all colloidal MCC products are marketed as MCC Gel.
A: Colloidal MCC is listed as E460(ii) — Powdered cellulose in the European Union under Regulation (EC) No 1333/2008, permitted in a wide range of food categories. In the United States, microcrystalline cellulose including colloidal forms holds GRAS (Generally Recognized as Safe) status under FDA 21 CFR 182.90. It is also monographed in USP/NF and Ph. Eur. for pharmaceutical use.According to the U.S. Food and Drug Administration (FDA), microcrystalline cellulose is classified as GRAS under 21 CFR 182.90.
Q: What is the typical use level of colloidal MCC in food applications? A: Usage levels vary by application. In ice cream, typical levels range from 0.1% to 0.5% by weight. In salad dressings and sauces, 0.3–1.0% is common. In pharmaceutical oral suspensions, concentrations of 0.5–2.5% are typical. Exact levels depend on the desired texture, the presence of other hydrocolloids, and processing conditions. Application trials are recommended to optimize dosage for each formulation.
Q: Can colloidal MCC be used with other stabilizers? A: Yes. Colloidal MCC is highly compatible with most hydrocolloids and stabilizers including carrageenan, locust bean gum, xanthan gum, pectin, CMC, and guar gum. In many applications, it is used in synergistic blends that allow manufacturers to achieve target texture and stability at lower total stabilizer cost. The MCC network provides suspension and body, while other hydrocolloids can contribute specific rheological properties.
Q: What is the difference between E460(i) and E460(ii)? A: E460(i) is microcrystalline cellulose (powder MCC) — a purified, partly depolymerized cellulose in crystalline powder form. E460(ii) is powdered cellulose, which includes colloidal MCC at finer particle sizes. Both fall under the broader E460 designation but have different particle characteristics and functional profiles in formulations.
Q: What is the difference between colloidal MCC and other hydrocolloids?
A: Unlike soluble hydrocolloids such as xanthan gum or pectin, colloidal MCC forms a particle-based three-dimensional network that provides superior suspension stability without significantly increasing viscosity.
Q: Why is colloidal MCC preferred in low-fat formulations?
A: Colloidal MCC mimics the mouthfeel of fat by creating a lubricating network structure, making it ideal for reduced-fat and calorie-controlled food products.
We are a specialized manufacturer of microcrystalline cellulose and colloidal MCC with over 15 years of production experience. Our products are widely used in food, pharmaceutical, and industrial formulations, with consistent export to more than 30 countries worldwide.
When it comes to sourcing colloidal MCC, reliability, compliance, and technical expertise are non-negotiable. Here is why leading food, pharmaceutical, and nutraceutical manufacturers consistently choose us as their preferred supplier.
With an annual production capacity exceeding 5,000 metric tons, we maintain consistent output to meet both large-scale industrial demands and flexible short-run requirements. Our vertically integrated manufacturing process — from raw cellulose sourcing to final spray-drying — ensures lead times of 2–4 weeks for standard grades and uninterrupted supply even during peak demand cycles. Safety stock levels are maintained at a minimum of 30 days’ rolling inventory to protect your production schedule.
Our colloidal MCC grades are fully compliant with major international regulatory frameworks, including:
| Regulation | Standard |
|---|---|
| US FDA | 21 CFR 182.90 / GRAS |
| European Union | EU 231/2012 (E460ii) |
| United States Pharmacopeia | USP-NF Microcrystalline Cellulose |
| Food Chemicals Codex | FCC Grade |
| Halal / Kosher | Certified |
Complete documentation packages — including Certificate of Analysis (CoA), Safety Data Sheet (SDS), allergen declarations, and origin certificates — are provided with every shipment, streamlining your regulatory submission process.
We offer a broad portfolio of viscosity grades ranging from 50 mPa·s up to 1,500 mPa·s, with the flexibility to develop tailored specifications based on your formulation requirements. Custom parameters include:
Prototype samples can typically be delivered within 3–5 business days for evaluation.
Our products are actively exported to over 30 countries across North America, Europe, Southeast Asia, the Middle East, and Oceania. We operate with established logistics partnerships supporting:
Our application technology team — with over 15 years of industry experience — provides end-to-end formulation assistance, including:
Ready to optimize your formulation? Contact our technical team today with your application details, and we will recommend the most suitable grade — backed by data, compliance, and supply security you can count on.
As a trusted colloidal microcrystalline cellulose supplier and manufacturer, we are committed to delivering consistent quality, stable viscosity performance, and reliable bulk supply for global food, pharmaceutical, and industrial applications. Our colloidal MCC solutions are widely used as stabilizers, suspending agents, and fat replacers in modern formulations.
Looking for a reliable bulk supplier of colloidal MCC with consistent quality, regulatory documentation, and technical support?
Visit our main [Microcrystalline Cellulose product page] for full specifications, available grades, minimum order quantities, and pricing — or speak directly with our formulation team about the right colloidal MCC grade for your application.
We supply food-grade, pharmaceutical-grade, and industrial-grade colloidal MCC to manufacturers across North America, Europe, and Asia.
评价
目前还没有评价